- Accueil
- Volume 15 (2011)
- numéro 3
- Large carrion beetles (Coleoptera, Silphidae) in Western Europe: a review
Visualisation(s): 7710 (44 ULiège)
Téléchargement(s): 1507 (1 ULiège)
Large carrion beetles (Coleoptera, Silphidae) in Western Europe: a review

Notes de la rédaction
Received on April 20, 2010; accepted on October 5, 2010
Résumé
Les grands coléoptères nécrophages (Coleoptera, Silphidae) en Europe occidentale : synthèse bibliographique. Cette synthèse bibliographique fait le point sur les Silphidae du Paléarctique Ouest et leur utilisation potentielle en entomologie forensique comme bioindicateurs postmortem. Peu d’études s’intéressent aux Silphidae dans un contexte forensique. Cependant, certaines espèces de Silphidae présentent les mêmes caractéristiques que certains Diptères que l’on utilise pour calculer un intervalle postmortem (IPM) et pourraient donc servir à calculer un IPM minimum plus étendu. La classification taxonomique et la distribution géographique des Silphidae sont décrites dans cette synthèse ainsi que leurs caractéristiques morphologiques et anatomiques, et ce pour les deux sous-familles de Silphidae. La biologie et l’écologie des Silphidae ont été synthétisées afin de mieux comprendre leur éventuelle valeur en tant qu’indicateur postmortem. Un chapitre spécifique met en évidence les différentes utilisations des Silphidae en entomologie forensique à l’heure actuelle.
Abstract
This review focuses on carrion beetles (Coleoptera, Silphidae) of the Western Palearctic and their potential use in forensic entomology as bioindicators. Few studies have looked at Silphidae in forensic context and investigations. However, some Silphidae present the desirable characteristics of some Diptera used in postmortem estimates and thus may extend the minimum postmortem interval (PMImin). We review here the taxonomy and distribution of Western Palearctic Silphidae. The anatomical and morphological characteristics of both subfamilies are described for adults and larvae. The biology and ecology of silphids are also summarized for Silphinae and Nicrophorinae. A specific chapter gives an overview of the current uses of Silphidae in forensic entomology as postmortem indicator.
Table des matières
1. Introduction
1Carrion beetles (Coleoptera, Silphidae) consist of a small group of Coleoptera counting less than 200 species that are worldwide spread (Sikes, 2008). Silphids perform vital ecosystem functions (Wolf et al., 2004); they promote the breakdown and recycling of organic matter into terrestrial ecosystems (Peck, 1990; Ratcliffe, 1996; Hastir et al., 2001; Kalinova et al., 2009). Most Silphids are carrion feeders (necrophagous species) but can also prey on other carrion inhabitants such as fly eggs or maggots and other small carrion beetles (necrophilous species) (Racliffe, 1996; Hastir et al., 2001; Sikes, 2005; Sikes, 2008). The “carrion” terminology is not adapted for all silphid species according to their ecological group, some species (Silphinae) are phytophagous or found in dung or fungi (Anderson et al., 1984; Sikes, 2008). Carrion beetles are also referenced as “large carrion beetles” contrary to other smaller carrion beetles such as Agyrtidae, Leiodidae (“small carrion beetles”) or Cholevidae (Peck, 1990; Ratcliffe, 1996; Peck, 2001; Sikes, 2008).
2Their feeding activities on carrion may also destroy some foci of infection of pathogenic bacteria (Peck, 1990). The necrophagous insects, including carrion beetles, have particular relationships with decomposing remains (vertebrate carcass) which constitute a rich ephemeral resource (Anderson et al., 1996; Grassberger et al., 2004; Carter et al., 2007). These specialized insects, including mainly Diptera and Coleoptera, are attracted to the cadaver that they colonize in a relative predictable sequence called the entomofaunal succession or insect succession (Megnin, 1894; Putman, 1983; Schoenly et al., 1987; Marchenko, 1988; Marchenko, 2001). Their study in a medico-legal context is a part of the forensic entomology (Amendt et al., 2004; Amendt et al., 2007). Many published reports or reviews are focused on Diptera pattern colonization and neglect Coleoptera succession (Kocarek, 2003; Matuszewski et al., 2008; Midgley et al., 2009). Carrion beetles have been referenced to as being a part of the entomofaunal colonization of a dead body but very few studies have looked at them in a forensic context. However, the use of beetles in forensic entomology can be relevant (Kulshrestha et al., 2001; Watson et al., 2005; Midgley et al., 2009; Midgley et al., 2010). Carrion beetles can provide information on postmortem colonization on remains and time since death (Smith, 1986; Haskell et al., 1997; Watson et al., 2005). This review focuses on Palearctic carrion beetles (Coleoptera, Silphidae) that are carrion feeder or associated with decomposing remains.
2. Taxonomy and distribution
3The family of Silphidae belongs to the superfamily of the Staphylinoidea and is divided into two subfamilies: the Nicrophorinae, called burying beetles or sexton beetles, and the Silphinae (Lawrence et al., 1982; Peck et al., 1993; Ratcliffe, 1996; Dobler et al., 2000; Sikes, 2005). Some taxonomists often include a third subfamily in the silphid beetles: the Agyrtinae (Madge, 1980; Hastir et al., 2001; Debreuil, 2003a; Debreuil, 2004a). However, recent phylogenetic analyses (Hansen, 1997; Newton, 1998; Dobler et al., 2000; Caterino et al., 2005) separate the Agyrtinae of other Silphidae and consider the Agyrtidae as a valid family into itself (Lawrence et al., 1982; Peck, 1990; Ratcliffe, 1996; Newton, 1997; Dobler et al., 2000; Caterino et al., 2005). The world fauna of Silphidae is currently composed of 183 species distributed in 15 genera (Ratcliffe, 1996; Peck, 2001; Sikes, 2005; Sikes, 2008). This family has a worldwide distribution, but is predominant in Holarctic regions (temperate regions) (Peck et al., 1985; Peck, 2001; Sikes, 2005). The Palearctic region is considered as the center of their distribution (Peck et al., 1985; Dobler et al., 2000). There are the most genera and the highest number of species of Silphidae in the Palearctic (Peck et al., 1985; Dobler et al., 2000). Carrion beetles are rare or absent in tropical regions because they are out-competed by ants, flies and vertebrates (Ratcliffe, 1996). Although, there are some Australian and Latin American endemic species (Diamesus, Ptomophila, Nicrophorus mexico) (Ratcliffe, 1996; Scott, 1998). Nicrophorinae are less widely distributed than Silphinae, being found in the temperate northern climate (Sikes, 2005; Sikes, 2008). Silphines seem to be more tolerant to warmer climate than the nicrophorines (Sikes, 2008). The subfamily of Silphinae has a greatest generic diversity (12 genera) than the Nicrophorinae (3 genera) (Sikes, 2005). In north Western Europe, there are 28 species of Silphidae: 11 species of Nicrophorinae and 17 species of Silphinae. Table 1 lists the species in Western Europe (Heinz, 1971; Hastir et al., 2001; Sikes et al., 2002; Debreuil, 2003a; Debreuil, 2003b; Debreuil, 2004a; Debreuil, 2004b; Debreuil, 2004c; Ružicka et al., 2004; Sikes, 2005). Among them, there are 22 species (11 Nicrophorus spp. and 11 Silphinae) that are carrion obligate or predacious species. There is only one genus of nicrophorine in the Western Palearctic: Nicrophorus. In the past, the spelling of this genus name varied from Nicrophorus to Necrophorus and back again to Nicrophorus, the valid genus name (Ratcliffe, 1996; Debreuil, 2004b), but it is not rare to see the wrong spelling in some publications.
3. Anatomic and morphological descriptions
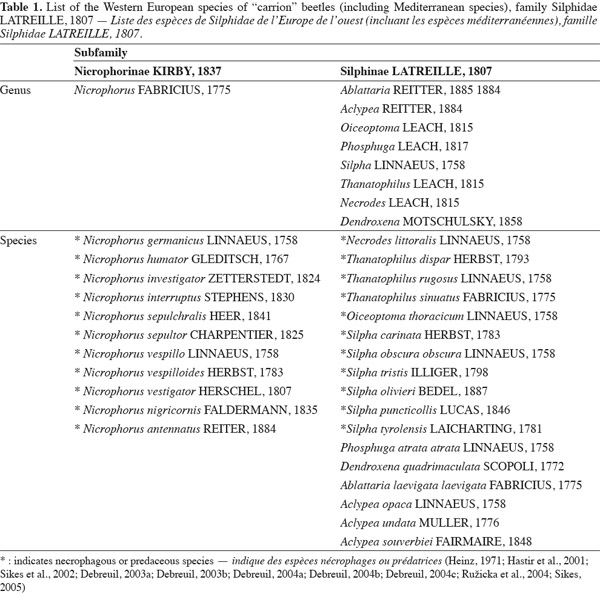
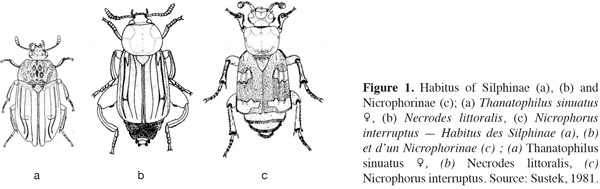
4Silphid beetles are usually medium to large in size (7 to 45 mm) (Peck, 1990; Ratcliffe, 1996; Hastir et al., 2001; Debreuil, 2003a; Sikes, 2008). Although, adults and larvae vary greatly in size and shape (Byrd et al., 2009). Adults have an ovate (Silphinae) to moderately elongate shape with protuberant eyes (Sikes, 2005) (Figure 1). They are flattened (Silphinae) or strongly convex (Ratcliffe, 1996). Silphids are often darkened or have distinctive red-orange-yellow markings on the elytra (Nicrophorus spp.) that may serve as warning coloration (Ratcliffe, 1996; Hastir et al., 2001). The elytra are often short and leave several abdominal segments exposed (1 or 5 abdominal segments among the subfamily). The elytra are punctuate and truncate in Necrodes (Silphinae) and Nicrophorinae, not truncate in the remaining Silphinae (Sikes, 2005) (Figure 2). The scutellum is often very large and the pronotum is enlarged (Peck, 1990; Sikes, 2005; Sikes, 2008).
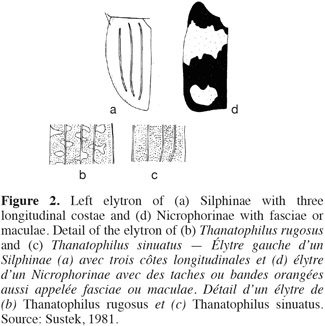
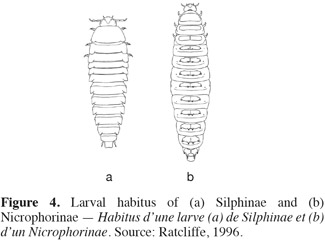
5The antennae are constituted by eleven segments and capitate (abruptly clubbed) for Nicrophorinae or clavate (gradually clubbed) for Silphinae (Hastir et al., 2001) (Figure 3). The antennae are widely spread and inserted on the lateral side of head. They have often microsetae covering only apical three segments (segments 9 to 11) (Hastir et al., 2001). The abdomen with sternite 2 is not visible between hind coxae but visible laterally of metacoxae (Sikes, 2008). The tarsi or terminal portion of each leg has five segments (tarsi 5-5-5) (Peck, 1990; Hastir et al., 2001). Silphid larvae are recognizable by the possession of a combination of: mandible without a molar lobe; maxilla with broad, apically cleft mala bearing setae on outer lobe; and usually a two-segmented articulated urogomphi (Newton, 1991). Each subfamily has a very distinctive habitus (Newton, 1991) (Figure 4).
3.1. Silphinae
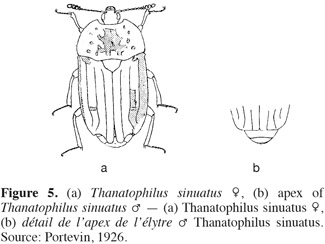
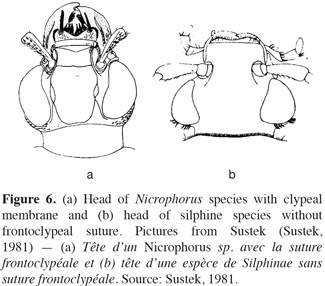
6Adults. Silphine species have often a darkened color and are dorsoventrally flattened (Figure 5). Their size is ranging from 8 to 25 mm (Debreuil, 2003a).The elytra have apices rounded or acute, not truncate or shortened (Peck, 1990; Ratcliffe, 1996). The elytra are usually costate or carinate (0-3 per elytron with 0 for the genus Ablattaria) (Peck, 1990; Debreuil, 2003a) but never striate (Figure 2a, 2b, 2c). The frontoclypeal suture is absent (Figure 6b) and the gular sutures are clearly separate, but strongly constricted medially (Peck, 1990; Sikes, 2005). The antennae have eleven well differentiated segments broaden gradually; particularly the last three or four segments (Figure 3b, 3c, 3d).
7Larvae. Silphine larvae are campodeiform with body surfaces heavily pigmented and sclerotized (Newton, 1991; Sikes, 2005; Sikes, 2008) (Figure 4a). The body length of mature larvae is ranging from 12 to 40 mm (Newton, 1991). On each side of the head, there are 6 pigmented stemmata (ocelli). The anal lobes bear numerous fine teeth (Sikes, 2005). The tergites are large, laterally produced; each tergite is usually with posterior angles attenuated (Anderson et al., 1985; Ratcliffe, 1996).
3.2. Nicrophorinae
8Adults. Nicrophorina species are darkened with red-orange markings (bands or spots), called fasciae or maculae, on elytra extending to the epipleura (Figure 1c), except for the species Nicrophorus humator and Nicrophorus germanicus which are completely darkened (black or brown) (Ratcliffe, 1996; Hastir et al., 2001; Sikes, 2008). Elytra have apices truncate (always smooth) and shortened exposing 3 or 4 abdominal segments (Peck, 1990; Ratcliffe, 1996) (Figures 1c and 2d). The frontoclypeal suture is present as a fine line (Peck, 1990; Sikes, 2005) (Figure 6a); the gular sutures are confluent posteriorly and reduce gula to a small triangular piece (Peck, 1990; Sikes, 2005). On the fifth dorsal segment, there is a pair of stridulatory files in both sexes which is used for communication (Lane et al., 1965; Peck, 1990; Ratcliffe, 1996; Sikes, 2008). The antennae are clubbed, the second antennal segment (pedicel) is often reduced and fewer differentiated than the scape (appearing 10-segmented) (Peck, 1990) (Figure 3a). The compact club is constituted by the last four antennal segments (Peck, 1990; Ratcliffe, 1996; Hastir et al., 2001). In most Nicrophorus species, there is a sexual dimorphism in tarsomeres. The nicrophorine males possess expanded segments of the protarsus (foretarsus) (Peck, 1990; Ratcliffe, 1996).
9Larvae. Nicrophorinae larvae are campodeiform or eruciform and body surfaces are lightly pigmented and unsclerotized with the exception of the head and legs (Newton, 1991; Ružicka, 1992; Sikes, 2005) (Figure 6b). The body length of mature larvae is ranging from 12 to 40 mm (Newton, 1991). The ventral surface is soft and creamy white (Anderson et al., 1985; Ružicka, 1992; Ratcliffe, 1996). On each side of the head, there is only one unpigmented stemma (Ratcliffe, 1996; Sikes, 2005). Contrary to Silphinae larvae, the anal lobes are without teeth (Newton, 1991; Sikes, 2005). The tergites are small; those on abdomen have 4 small spines (Anderson et al., 1985; Ratcliffe, 1996). For more detailed information about the morphology of larvae of Nicrophorus, Ružicka (1992) describes the immature stages of the following European species: N. vespillo, N. vespilloides, N. humator, N. investigator, N. (fossor) interruptus.
4. Ecology and biology
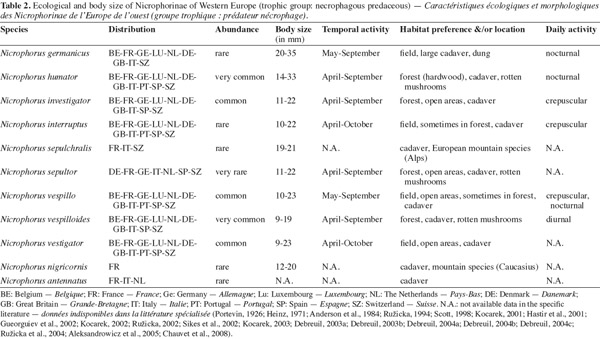
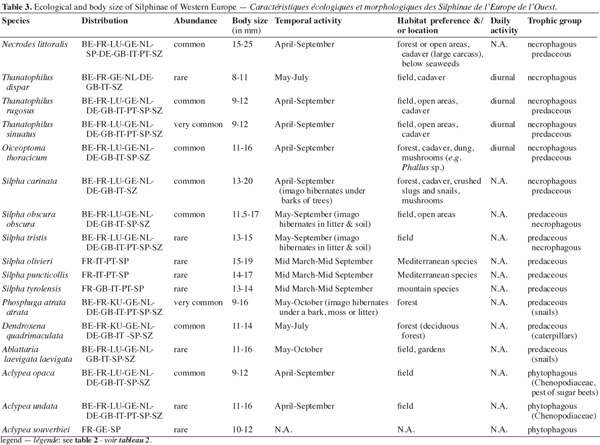
10Carrion beetles (Coleoptera, Silphidae) perform vital ecosystem functions (Wolf et al., 2004); they promote the breakdown and recycling of organic matter into terrestrial ecosystems (Ratcliffe, 1996; Hastir et al., 2001; Kalinova et al., 2009). Tables 2 and 3 list the different ecological characteristics of Nicrophorinae and Silphinae Western European species based on the relevant literature (Portevin, 1926; Heinz, 1971; Anderson et al., 1984; Ružicka, 1994; Scott, 1998; Kocarek, 2001; Hastir et al., 2001; Gueorguiev et al., 2002; Kocarek, 2002; Ružicka, 2002; Sikes et al., 2002; Kocarek, 2003; Debreuil, 2003a; Debreuil, 2003b; Debreuil, 2004a; Debreuil, 2004b; Debreuil, 2004c; Ružicka et al., 2004; Aleksandrowicz et al., 2005; Chauvet et al., 2008). Silphidae are mainly carrion feeder (necrophagous species) or prey on other carrion inhabitants such as fly eggs or maggots and other carrion beetles (necrophilous species) (Racliffe, 1996; Hastir et al., 2001; Sikes, 2005; Sikes, 2008). There are some species of Silphinae that feed on soil invertebrates (snails, caterpillars or slugs predators: Silpha spp., Dendroxena spp.) or are phytophagous species (e.g. Aclypea spp.) (Sikes, 2005; Ikeda et al., 2007; Ikeda et al., 2008). Some Silphidae may be attracted by decaying fungi (e.g. Phallus impudicus), dung or rotten plant (Ratcliffe, 1996; Hastir et al., 2001; Sikes, 2005). Silphidae have reduced interspecific competition in partitioning species (niche differentiation) (Anderson, 1982; Peck, 1990; Ohkawara et al., 1998; Hocking et al., 2007): they have different temporal activities; some species are more active during spring whereas other species are active in summer, few species are active during autumn (Ružicka, 1994; Scott, 1998; Kocarek, 2001). In carrion beetles communities, niche differentiation can occur along dimensions of season, habitat (biotope) and carcass size (Scott, 1998; Hocking et al., 2007). The daily activity is also different, Silphidae are primarily nocturnal insect but some species are diurnal (e.g. Thanatophilus spp.) or crepuscular (Ohkawara et al., 1998; Scott, 1998; Kocarek, 2001). Habitat preferences are also different; some species are subservient to forest biotope (e.g. O. thoracica, N. vespilloides), whereas other species prefer open habitats (field/meadow species) (Scott, 1998; Kocarek, 2001). The type of carrion (age and carcass size) that they use is an important parameter (Anderson, 1982; Peck, 1990; Scott, 1998). Silphinae tend to use preferentially large vertebrate carcasses whereas Nicrophorinae prefer small carcasses (Anderson, 1982; Peck, 1990; Eggert et al., 1992). Both subfamilies have different reproductive strategy. Nicrophorinae have a surprising behavior for insects: the biparental care of their offspring (Pukowski, 1933; Anderson, 1982; Scott, 1998; Smiseth et al., 2006). This is the highest level of sociability attained in the Coleoptera (Milne et al., 1976; Ratcliffe, 1996). At contrary, Silphinae show no parental care (Ratcliffe, 1996). Silphidae have particular relationships with nematodes and mites (phoresy) (Springett, 1968; Richter, 1993; Ratcliffe, 1996; Sikes, 2008). These relationships, poorly known, could be mutualism, commensalism or parasitism (Sikes, 2008).
4.1. Silphinae
11Contrary to the Nicrophorinae, little is known about the biology and ecology of the Silphinae (Ratcliffe, 1996; Hoback et al., 2004; Ikeda et al., 2007). Concerning necrophagous silphine species, females are semelparous and lay their eggs in or on the soil around large vertebrate carcasses and provide no care of their offspring (Sikes, 2005; Ikeda et al., 2008). Silphine appear usually on large carcasses (> 300 g) (Peck, 1990; Sikes, 2005) because these provide sufficient food resource for the great number of beetles that may be present (Anderson, 1982; Watson et al., 2005). Eggs hatch in 4-5 days and silphine larvae feed on carrion remains (Anderson, 1982). They may also compete with nicrophorine species for small vertebrate carcasses that they use for feed but not for reproduction and larval development (Bishop, 2001; Hoback et al., 2004). Silphinae colonize a carcass during the early or mid-stage of decay and thus compete with flies (Diptera) for the food resource (Payne, 1965; Anderson, 1982). Contrary to Nicrophorinae, there are some flightless silphine species or some flight-dimorphic species (Ikeda et al., 2007; Ikeda et al., 2008).
4.2. Nicrophorinae
12Many studies on burying beetles behavior have been published since the pioneer work of Pukowski (Pukowski, 1933; Milne et al., 1976; Sikes, 2005). More than 150 behavioral ecology studies on the Nicrophorus spp. were conducted in the past 25 years (Sikes, 2008). The reviews of Milne et al. (1976), Ratcliffe (1996), Scott (1998) or Sikes (2005; 2008) provide detailed information about the nicrophorine (or Silphidae) ecology. Burying beetles specialized on carrion (necrophagous and necrophilous species) are subsocial (Pukowski, 1933; Milne et al., 1976; Trumbo et al., 1993; Ohkawara et al., 1998; Scott, 1998). Nicrophorus species use small vertebrate carcasses (< 300 g, usually < 100 g) such as rodents or birds that they bury and prepare for rearing offspring (Pukowski, 1933; Milne et al., 1976; Trumbo, 1990b; Scott, 1998; Smith et al., 2001; Sikes, 2005; Sikes, 2008). When the carcass is found by a single pair of male and female, they search a suitable spot for burial that is usually completed in 5 to 8 hours during the night (Ratcliffe, 1996; Scott, 1998). Carcasses are often located by several individuals of both sexes (Pukowski, 1933; Müller et al., 1998; Steiger et al., 2009). In this case, fighting occurs between individuals for the ownership of the carcass by a single male-female pair (Pukowski, 1933; Müller et al., 1998; Steiger et al., 2009). Searching behavior is guided by olfaction; burying beetles have sensitive chemosensors located on their antennae adapted to detect the smell of a recently dead animal (Shubeck, 1975; Bartlett, 1987; Peck, 1990; Ratcliffe, 1996; Kalinova et al., 2009; Steiger et al., 2009). If a male discovers a suitable carcass for reproduction, it emits a sexual pheromone to attract the female (Eggert et al., 1989; Eggert, 1992; Ohkawara et al., 1998). After the burial [10-20 cm depth (Hastir et al., 2001)], Nicrophorine removes the skin (fur or feathers) and the remains are fashioned into a compact ball. Then, they inoculate the carrion ball with oral and anal secretions that have antimicrobial properties to delay the decomposition process (Ratcliffe, 1996; Eggert et al., 1997; Scott, 1998; Hoback et al., 2004; Rozen et al., 2008; Cotter et al., 2010) and remove also fungi (Scott, 1998). The female makes a chamber above the carrion ball in which it lays 10-50 eggs. Both parents regurgitate food in this crypt for feeding their larvae. Larvae may also feed directly on the surface of the carrion ball (Ratcliffe, 1996; Ohkawara et al., 1998; Sikes, 2008). The larvae receive parental care during their entire development (Ratcliffe, 1996; Scott, 1998). Parents provide extensive care: they feed their offspring, they protect them from predators and intruding burying beetles (inter- and intraspecific competitions) and they maintain a pathogen free nest with preservative secretions (Ratcliffe, 1996; Scott, 1998; Smiseth et al., 2006). Female stays on the crypt until complete larval development (1-4 weeks), whereas the male abandons the brood a few days earlier (Trumbo, 1991; Müller et al., 1998). If the brood is too large for a successful development, adults may regulate brood size by selective cannibalism. They kill smaller larvae during the first 24 h after hatching (Trumbo, 1990a; Ratcliffe, 1996). After one week, the larvae have consumed the entire carrion ball and pupate in the nearby soil during two weeks (Ratcliffe, 1996; Ohkawara et al., 1998). Then, adults emerge and the overwintering occurs in the adult stage or in the pre-pupal stage for some species (Ratcliffe, 1996; Ohkawara et al., 1998). Sometimes, nicrophorine may colonize large carcass (too large to bury) and several male-female pairs breed communally their larvae in a subsocial fashion: cooperative breeding (Pukowski, 1933; Eggert et al., 1992; Ratcliffle, 1996; Scott, 1998).
5. The utility of carrion beetles in forensic entomology
13Most forensic researches have focused on flies while beetles have been neglected (Midgley et al., 2009; Midgley et al., 2010). When a corpse colonized by insects is found, two situations could be considered (Amendt et al., 2007; Lefebvre et al., 2009). In the first situation, which is the most frequent case in forensic investigations, insects are pioneer species and the minimum postmortem interval (PMI) is estimated with the age of the oldest specimens found on the death scene, principally blowflies (Amendt et al., 2007; Lefebvre et al., 2009). In the second situation, later necrophagous species colonize the corpse with a delay, often after the departure of pioneer species. The estimation of the PMI is only possible by analyzing the chronological succession (Amendt et al., 2007; Lefebvre et al., 2009). A frequent objection against the use of Coleoptera in forensic investigations is the fact that flies (pioneer species) locate corpses faster than beetles (later necrophagous species). Thus, the minimum postmortem interval estimates are more accurate with Diptera, especially with the families of Calliphoridae and Sarcophagidae (Smith, 1986; Midgley et al., 2010). However, recent researches have shown that some Silphidae (e.g. Thanatophilus micans FABRICIUS) can locate a corpse within 24 h and their larvae have been observed soon after death, during the early stage of decomposition (Midgley et al., 2009; Midgley et al., 2010). This implies that some carrion beetles have the same forensic interesting characteristics than carrion flies and can be considered as pioneer species. In this case, some species of Coleoptera can be used as reliable forensic indicators such as blowflies (Midgley et al., 2009; Midgley et al., 2010). However, there are no available information about early postmortem colonization by European carrion beetles such as in South Africa with T. micans. Some recent publications (Matuszewski et al., 2008; Matuszewski et al., 2010) associate the silphid activity on carcasses during the active decay stage, primarily for Silphinae (N. littoralis, Thanatophilus spp.). The most important application of insects in forensic investigations is the estimation of the minimum PMI (Greenberg, 1991; Amendt et al., 2004; Amendt et al., 2007). These minimum PMI estimates are primarily based on the duration of immature stages of Diptera (development models) (Amendt et al., 2007). Contrary to flies, there are few studies on the rates of development of Coleoptera with forensic interest (Midgley et al., 2009; Midgley et al., 2010). For example, Midgley et al. (2009) studied the development of T. micans at ten constant temperatures. They established a robust statistical model of development for this common African species. Currently, there is no development model (“size-at-age data”) for forensically relevant European silphids. However, research on development of Coleoptera with a forensic interest can be a useful tool for medico-legal entomologists (Midgley et al., 2010). In addition, carrion beetles have generally a longer life cycle than forensic Diptera (Midgley et al., 2009; Midgley et al., 2010). They can colonize a corpse during later decay stages when many maggots have already left the corpses (Kocarek, 2003; Matuszewski et al., 2008; Midgley et al., 2010). The PMI estimates can be established by analyzing the arthropod community present on a corpse including many Coleoptera during the later stage of decomposition (Smith, 1986). However, the biology and ecology of most forensically relevant species of Coleoptera are unknown (Midgley et al., 2010). To increase the accuracy and the validity of the PMI estimates based on ecological successions, there is a necessity to generate data on insect succession and insect seasonal activity on carrion in specific geographic regions and various biotopes within these regions (Catts et al., 1992; Amendt et al., 2004; Sharanowski et al., 2008; Lefebvre et al., 2009). All carrion beetles do not have the same forensic interest; species of Silphinae seem to have a more important value as forensic bioindicators (Watson et al., 2005; Matuszewski et al., 2010). Indeed they have ecological preferences for small vertebrate carcasses, while Nicrophorinae present less interest in forensic entomology (Watson et al., 2005). However, Nicrophorus spp. could be frequently found on human corpses, including in houses (Chauvet et al., 2008). This is an inventory extracted from 700 real forensic cases that occurred during 15 years in France. Midgley et al. (2010) suggest to focus on the biology of both Silphinae (Silpha and Thanatophilus). While Matuszewski et al. (2010) highlight the forensic usefulness of the following silphine species: Necrodes littoralis (larvae and adults), Thanatophilus spp. (larvae and adults) and O. thoracica (larvae). In some cases, necrophagous beetles can also provide information on the presence of drugs or poisons by bioaccumulation (entomotoxicology) (Bourel et al., 2001; Introna et al., 2001; Carvalho, 2010). Adults, larvae or beetle remain such as exuviae, puparial cases or fecal material of Coleopterans may be used for toxicological analysis when conventional toxicological samples (blood, urine, internal organs) are not available (Miller et al., 1994; Bourel et al., 2001; Introna et al., 2001; Carvalho, 2010).
6. Conclusion
14The potential uses of European carrion beetles as bioindicators in forensic entomology are obvious. Silphids and principally burying beetles (Nicrophorus spp.) are widely studied in various contexts including biology and ecology (Ratcliffe, 1996; Scott, 1998). Indeed, carrion beetles are poorly studied in a forensic context (Midgley et al., 2010). Nevertheless, their use in forensic investigations can be relevant (Watson et al., 2005; Midgley et al., 2009; Midgley et al., 2010). Are there some European carrion beetles with forensically interesting characteristics? Before creating development model for Paleartic Silphidae of forensic value, forensic entomologists need to increase data on carrion beetle’s ecology and insect succession.
15Acknowledgements
16Jessica Dekeirsschieter is financially supported by a PhD grant from the Fonds pour la Formation à la Recherche dans l’Industrie et l’Agriculture (F.R.I.A), Belgium.
Bibliographie
Aleksandrowicz O. & Komosinski K., 2005. On the fauna of carrion beetles (Coleoptera, Silphidae) of Mazurian lakeland (north-eastern Poland). In: Sklodowski J., Huruk S., Bersevskis A. & Tarasiuk S., eds. Protection of Coleoptera in the Baltic Sea region. Warsaw: Warsaw Agricultural University Press, 147-153.
Amendt J., Krettek R. & Zehner R., 2004. Forensic entomology. Naturwissenschaften, 91, 51-65.
Amendt J. et al., 2007. Best practice in forensic entomology. Standards and guidelines. Int. J. Legal Med., 121, 90-104.
Anderson R.S., 1982. Resource partitioning in the carrion beetle (Coleoptera: Silphidae) fauna of southern Ontario: ecological and evolutionary considerations. Can. J. Zool., 60, 1314-1325.
Anderson R.S. & Peck S.B., 1984. Binomics of neartic species of Aclypea Reitter: phytophagous “carrion” beetles (Coleoptera: Silphidae). Pan-Pacific Entomologist, 60(3), 248-255.
Anderson R.S. & Peck S.B., 1985. The carrion beetles of Canada and Alaska (Coleoptera: Silphidae and Agyrtidae). The insects and arachnids of Canada. Part 13. Ottawa, ON, Canada: Biosystematics Research Institute.
Anderson G.S. & VanLaerhoven S.L., 1996. Initial studies on insect succession on carrion in Southwestern British Columbia. J. Forensic Sci., 41, 617-625.
Bartlett J., 1987. Evidence for a sex attractant in burying beetles. Ecol. Entomol., 12, 471-472.
Bishop A.A., 2001. Niche segregation, carcass preparation, and the effects of land management on the ecology of carrion beetles (Silphidae) in Kearney County, Nebraska. M.S. thesis: University of Nebraska at Kearney (USA).
Bourel B. et al., 2001. Morphine extraction in necrophagous insects remains for determining ante-mortem opiate intoxication. Forensic Sci. Int., 120, 127-131.
Byrd J.H. & Castner J.L., 2009. Insects of forensic importance. In: Byrd J.H. & Castner J.L., eds. Forensic entomology. The utility of arthropods in legal investigations. Boca Raton, FL, USA: CRC Press, 39-126.
Carter D.O., Yellowlees D. & Tibbett M., 2007. Cadaver decomposition in terrestrial ecosystems. Naturwissenschaften, 94, 12-24.
Carvalho L.M.P., 2010. Toxicology and forensic entomology. In: Amendt J., Campobasso C.P., Goff M.L. & Grassberger M., eds. Current concepts in forensic entomology. Dordrecht, The Netherlands; Heidelberg, Deutschland; London; New York, USA: Springer, 163-178.
Caterino M.S., Hunt T. & Vogler A.P., 2005. On the constitution and phylogeny of Staphyliniformia (Insecta: Coleoptera). Mol. Phylogenet. Evol., 34, 655-672.
Catts E.P. & Goff M.L., 1992. Forensic entomology in criminal investigations. Annu. Rev. Entomol., 37, 253-272.
Chauvet B. et al., 2008. Répartition des Nicrophorus F., 1775 récoltés sur des cadavres humains (Coleoptera, Silphidae). L’Entomologiste, 64(1), 15-19.
Cotter S.C. & Kilner R.M., 2010. Sexual division of antibacterial resource defense in breeding burying beetles, Nicrophorus vespilloides. J. Anim. Ecol., 79, 35-43.
Debreuil M., 2003a. Contribution à la connaissance de la famille des Silphidae Latreille, 1807 (Coleoptera Staphylinoidea). 1ère partie. Rutilans, 6(2), 29-37.
Debreuil M., 2003b. Contribution à la connaissance de la famille des Silphidae Latreille, 1807 (Coleoptera Staphylinoidea). 2e partie. Rutilans, 6(3), 59-63.
Debreuil M., 2004a. Contribution à la connaissance de la famille des Silphidae Latreille, 1807 (Coleoptera Staphylinoidea). 3e partie. Rutilans, 7(1), 17-25.
Debreuil M., 2004b. Contribution à la connaissance de la famille des Silphidae Latreille, 1807 (Coleoptera Staphylinoidea). 4e partie. Rutilans, 7(2), 31-37.
Debreuil M., 2004c. Contribution à la connaissance de la famille des Silphidae Latreille, 1807 (Coleoptera Staphylinoidea). 5e partie. Rutilans, 7(3), 73-78.
Dobler S. & Müller J.K., 2000. Resolving phylogeny at the family level by mitochondrial cytochrome oxidase sequences: phylogeny of carrion beetles (Coleoptera, Silphidae). Mol. Phylogenet. Evol., 15(3), 390-402.
Eggert A.-K., 1992. Alternative male mate-finding tactics in burying beetles. Behav. Ecol., 3, 243-254.
Eggert A.-K. & Müller J.K., 1989. Pheromone-mediated attraction in burying beetles. Ecol. Entomol., 14, 235-237.
Eggert A.-K. & Müller J.K., 1992. Joint breeding in female burying beetles. Behav. Ecol. Sociobiol., 31, 237-242.
Eggert A.-K. & Muller J.K., 1997. Biparental care and social evolution in burying beetles: lessons from the larder. In: Choe J. & Crespi B., eds. The evolution of social behavior in insects and arachnids. Cambridge, UK: Cambridge University Press, 216-236.
Grassberger M. & Frank C., 2004. Initial study of arthropod succession on pig carrion in a central European urban habitat. J. Med. Entomol., 41, 511-523.
Greenberg B., 1991. Flies as forensic indicators. J. Med. Entomol., 28, 565-577.
Gueorguiev B. & Ružicka J., 2002. Check list of Bulgarian carrion beetles (Coleoptera: Silphidae). Historia naturalis bulgarica, 15, 89-112.
Hansen M., 1997. Phylogeny and classification of the staphyliniform beetle families (Coleoptera). Biol. Skrifter, 48, 1-339.
Haskell N., Hall R., Cervenka V. & Clark M., 1997. On the body: insects’ life stage presence and their postmortem artifacts. In: Haglund W.D. & Sorg M.H., eds. Forensic taphonomy: the postmortem fate of human remains. Boca Raton, FL, USA: CRC Press, 415-445.
Hastir P. & Gaspar C., 2001. Diagnose d’une famille de fossoyeurs : les Silphidae. Notes fauniques de Gembloux, 44, 13-25.
Heinz F., 1971. Familie: Silphidae (aaskäfer). In: Heinz F., Harde K.W. & Loshe G.A., eds. Die Käfer Mitteleuropas, Band 3. Adephaga 2, Palpicornia, Histeroidea, Staphylinoidea 1. Krefeld, Germany: Goecke & Evers Verlag, 190-201.
Hoback W.W. et al., 2004. Differences among antimicrobial properties of carrion beetle secretions reflect phylogeny and ecology. J. Chem. Ecol., 30(4), 719-729.
Hocking M.D., Darimont C.T., Christie K.S. & Reimchen T.E., 2007. Niche variation in burying beetles (Nicrophorus spp.) associated with marine and terrestrial carrion. Can. J. Zool., 85, 437-442.
Ikeda H., Kubota K. & Abe T.K.T., 2007. Flight capabilities and feeding habits of silphine beetles: are flightless species really “carrion beetles”? Ecol. Res., 22, 237-241.
Ikeda H., Kagaya T., Kubota K. & Abe T., 2008. Evolutionary relationships among food habit, loss of flight, and reproductive traits: life-history evolution in the Silphinae (Coleoptera: Silphidae). Evolution, 62(8), 2065-2079.
Introna F., Campobasso C.P. & Goff M.L., 2001. Entomotoxicology. Forensic Sci. Int., 120, 42-47.
Kalinova B., Podskalska H., Ružicka J. & Hoskovee M., 2009. Irresistible bouquet of death. How are burying beetles (Coleoptera: Silphidae: Nicrophorus) attracted by carcasses. Naturwissenschaften, 96, 889-899.
Kocarek P., 2001. Diurnal activity rhythms and niche differentiation in a carrion beetle assemblage (Coleoptera: Silphidae) in Opava, the Czech Republic. Biol. Rhythm Res., 32(4), 431-438.
Kocarek P., 2002. Diel activity patterns of carrion-visiting Coleoptera studied by time-sorting pitfall traps. Biologia Bratislava, 57(2), 199-211.
Kocarek P., 2003. Decomposition and coleoptera succession on exposed carrion of small mammal in Opava, the Czech Republic. Eur. J. Soil Biol., 39, 31-45.
Kulshrestha P. & Satpathy D.K., 2001. Use of beetles in forensic entomology. Forensic Sci. Int., 120, 15-17.
Lane C. & Rothschild M., 1965. A case of Müllerian mimicry of sound. Proc. R. Entomol. Soc. Lond., 40, 156-158.
Lawrence J.F. & Newton A.F.J., 1982. Evolution and classification of beetles. Annu. Rev. Ecol. Syst., 13, 261-290.
Lefebvre F. & Gaudry E., 2009. Forensic entomology: a new hypothesis for the chronological succession pattern of necrophagous insect on human corpses. Ann. Soc. Entomol. Fr., 45, 377-392.
Madge R.B., 1980. A catalogue of type-species in the family Silphidae (Coleoptera). Entomol. Scand., 11, 353-362.
Marchenko M.I., 1988. Medico-legal relevance of cadaver entomaufauna for the determination of the time since death. Acta Med. Leg. Soc., 38, 257-302.
Marchenko M.I., 2001. Medico-legal relevance of cadaver entomofaune for the determination of time of death. Forensic Sci. Int., 120, 89-109.
Matuszewski S., Bejerlein D., Konwerski S. & Szpila K., 2008. An initial study of insect succession and carrion decomposition in various forest habitats of Central Europe. Forensic Sci. Int., 180, 61-69.
Matuszewski S., Bejerlein D., Konwerski S. & Szpila K., 2010. Insect succession and carrion decomposition in selected forests of Central Europe. Part 2: Composition and residency patterns of carrion fauna. Forensic Sci. Int., 195, 42-51.
Megnin J.P., 1894. La faune des cadavres, application de l’entomologie à la médecine légale. Paris : L’Encyclopédie Scientifique des Aide-mémoire.
Midgley J.M. & Villet M.H., 2009. Development of Thanatophilus micans (Fabricius 1794) (Coleoptera Silphidae) at constant temperatures. Int. J. Legal Med., 123, 285-292.
Midgley J.M., Richards C.S. & Villet M.H., 2010. The utility of Coleoptera in forensic investigations. In: Amendt J., Goff M.L., Campobasso C.P. & Grassberger M., eds. Current concepts in forensic entomology. Dordrecht, The Netherlands: Springer, 57-68.
Miller M.L. et al., 1994. Isolation of amitriptyline and nortriptyline from fly puparia (Phoridae) and beetle exuviae (Dermestidae) associated with mummified human remains. J. Forensic Sci., 39, 1305-1313.
Milne L.J. & Milne M., 1976. The social behavior of burying beetles. Sci. Am., 235, 84-89.
Müller J.K., Eggert A.-K. & Sakaluk S.K., 1998. Carcass maintenance and biparental brood care in burying beetles: are males redundant? Ecol. Entomol., 23, 195-200.
Newton A.F.J., 1991. Silphidae. In: Stehr F.W., ed. Immature insects. Vol. 2. Dubuque, IA, USA: Kendall/Hunt, 339-341.
Newton A.F.J., 1997. Review of Agyrtidae (Coleoptera), with a new genus and species of New Zealand. Ann. Zool. Warszawa, 47, 111-156.
Newton A.F.J., 1998. Phylogenetic problems, current classification, and genetic catalog of world Leiodidae (including Cholevidae). In: Giachino P.M. & Peck S.B., eds. Phylogeny and evolution of subterranean and endogean Cholevidae (= Leiodidae, Cholevinae), Proceedings of 20th International Congress of Entomology, 1996, Firenze, Italy. Torino, Italy: Atti Museo Regionale di Scienze Naturali, 41-178.
Ohkawara K., Suzuki S. & Katakura H., 1998. Competitive interaction and niche differenciation among burying beetles (Silphidae, Nicrophorus) in Northern Japan. Entomol. Sci., 1(4), 551-559.
Payne J., 1965. A summer carrion study of the baby pig Sus scrofa Linnaeus. Ecology, 46, 592-602.
Peck S.B., 1990. Insecta: Coleoptera Silphidae and the associated families Agyrtidae and Leiodidae. In: Dindal D.L., ed. Soil biology guide. New York, USA: John Wiley & Sons, 1113-1136.
Peck S.B., 2001. Review of the carrion beetles of Australia and New Guinea (Coleoptera: Silphidae). Aust. J. Entomol., 40, 93-101.
Peck S.B. & Anderson R.S., 1985. Taxonomy, phylogeny and biogeography of the carrion beetles of Latin America (Coleoptera: Silphidae). Quaestiones Entomologicae, 21, 247-317.
Peck S.B. & Miller S.E., 1993. A catalog of the Coleoptera of America North of Mexico. Family Silphidae. Washington, DC, USA: USDA, 47-81.
Portevin M.G., 1926. Les grands nécrophages du globe, Silphini-Necrodini-Necrophorini. Encyclopédie Entomologique (A). Vol. 6. Paris: Paul Lechevalier.
Pukowski E., 1933. Ökologische Untersuchungen an Necrophorus F. Z. Morphol. Ökologie Tiere, 27, 518-586.
Putman R.J., 1983. Carrion and dung: studies in biology. London: Edward Arnold, 59.
Ratcliffe B.C., 1996. The carrion beetles (Coleoptera: Silphidae) of Nebraska. Bulletin 13. Lincoln, NE, USA: University of Nebraska State Museum.
Richter S., 1993. Phoretic association between the dauerjuveniles of Rhabditis stammeri (Rhabditidae) and life history stages of the burying beetle Nicrophorus vespilloides (Coleopetra: Silphidae). Nematologica, 39, 346-355.
Rozen D.E., Engelmoer D.J.P. & Smiseth P.T., 2008. Antimicrobial strategies in burying beetles breeding on carrion. Proc. Natl Acad. Sci., 105(46), 17890-17895.
Ružicka J., 1992. The immature stages of central European species of Nicrophorus (Coleoptera, Silphidae). Acta Entomol. Bohemoslovaca, 89, 113-135.
Ružicka J., 1994. Seasonal activity and habitat associations of Silphidae and Leiodidae: Cholevinae (Coleoptera) in Central Bohemia. Acta Soc. Zool. Bohemia, 58, 67-78.
Ružicka J., 2002. Taxonomic and nomenclatorial notes on Palaearctic Silphinae (Coleoptera: Silphidae). Acta. Soc. Zool. Bohemia, 66, 303-320.
Ružicka J., Hava J. & Schneider J., 2004. Revision of Palearctic and Oriental Oiceoptoma (Coleoptera: Silphidae). Acta Soc. Zool. Bohemia, 68, 30-51.
Ružicka J. & Schneider J., 2004. Family Silphidae Latreille, 1807. In: Löbl I. & Smetana A., eds. Catalogue of Palaearctic Coleoptera. Vol. 2: Hydrophiloidea - Histeroidea - Staphylinoidea. Steensrup, Denmark: Apollo Books, 229-237.
Schoenly K. & Reid W., 1987. Dynamic or heterotrophic succession in carrion arthropod assemblages: discrete seres or a continuum of change. Oecology, 73, 192-202.
Scott M.P., 1998. The ecology and behaviour of burying beetles. Annu. Rev. Entomol., 43, 595-618.
Sharanowski B.J., Walker E.G. & Anderson G.S., 2008. Insect succession and decomposition patterns on shaded and sunlit carrion in Saskatchewan in three different seasons. Forensic Sci. Int., 179, 219-240.
Shubeck P.P., 1975. Do carrion beetles use sight, as an aid to olfaction, in locating carrion? William L. Hutcheson Memorial For. Bull., 3, 36-39.
Sikes D.S., 2005. Silphidae Latreille, 1807. In: Kristensen N. P. & Beutel R.G., eds. Handbook of Zoology. Vol. 4: Arthropoda: Insecta. Berlin, Germany: Waler de Gruyter, 288-296.
Sikes D.S., 2008. Carrion beetles (Coleoptera: Silphidae). In: Capinera J.L., ed. Encyclopedia of entomology. Dordrecht, The Netherlands: Springer, 749-757.
Sikes D.S., Madge R.B. & Newton A.F., 2002. A catalog of the Nicrophorinae (Coleoptera: Silphidae) of the world. Zootaxa, 65, 1-304.
Smiseth P.T., Musa S. & Moore A.J., 2006. Negotiation between parents: does the timing of mate loss affect female compensation in Nicrophorus vespilloides. Behavior, 143, 293-301.
Smith K.G.V., 1986. A manual of forensic entomology. London: The Trustees of the British Museum, Natural History.
Smith R.J. & Merrick M.J., 2001. Resource availability and population dynamics of Nicrophorus investigator, an obligate carrion breeder. Ecol. Entomol., 26, 173-180.
Springett B.P., 1968. Aspects of the relationship between burying beetles, Necrophorus spp., and the mite, Poecilochirus necrophori Vitz. J. Anim. Ecol., 37, 417-424.
Steiger A., Whitlow S., Peschke K. & Müller J.K., 2009. Surface chemicals inform about sex and breeding status in the biparental burying beetle Nicrophorus vespilloides. Ethology, 115, 178-185.
Šustek Z., 1981. Key to identification of insects: carrion beetles of Czechoslovakia (Coleoptera, Silphidae). Zprávy Československé Společnosti Entomologické při ČSAV, Klíče k určování hmyzu, 2, 1-47.
Trumbo S.T., 1990a. Reproductive benefits of infanticide in a biparental burying beetle Nicrophorus orbicollis. Behav. Ecol. Sociobiol., 27, 267-273.
Trumbo S.T., 1990b. Interference competition among burying beetles (Silphidae, Nicrophorus). Ecol. Entomol., 15, 347-355.
Trumbo S.T., 1991. Reproductive benefits and the duration of paternal care in a biparental burying beetle, Necrophorus orbicollis. Behavior, 117, 82-105.
Trumbo S.T. & Fiore A.J., 1993. Interspecific competition and the evolution of communal breeding in burying beetles. Am. Midland Nat., 131(1), 169-174.
Watson E.J. & Carlton C.E., 2005. Succession of forensically significant carrion beetle larvae on large carcasses (Coleoptera: Silphidae). Southeastern Nat., 4(2), 335-346.
Wolf J. & Gibbs J., 2004. Silphids in urban forests: diversity and function. Urban Ecosyst., 7, 371-384.
Pour citer cet article
A propos de : Jessica Dekeirsschieter
Univ. Liege - Gembloux Agro-Bio Tech. Department of Functional and Evolutionary Entomology. Passage des Déportés, 2. B-5030 Gembloux (Belgium). E-mail: jdekeirsschieter@ulg.ac.be, entomologie.gembloux@ulg.ac.be
A propos de : François Verheggen
Univ. Liege - Gembloux Agro-Bio Tech. Department of Functional and Evolutionary Entomology. Passage des Déportés, 2. B-5030 Gembloux (Belgium).
A propos de : Georges Lognay
Univ. Liege - Gembloux Agro-Bio Tech. Department of Analysis Quality and Risks. Laboratory of Analytical Chemistry. Passage des Déportés, 2. B-5030 Gembloux (Belgium).
A propos de : Éric Haubruge
Univ. Liege - Gembloux Agro-Bio Tech. Department of Functional and Evolutionary Entomology. Passage des Déportés, 2. B-5030 Gembloux (Belgium).





