- Accueil
- volume 16 (2012)
- numéro 3
- Involvement of phenolic compounds in the susceptibility of bananas to crown rot. A review
Visualisation(s): 18483 (208 ULiège)
Téléchargement(s): 460 (5 ULiège)
Involvement of phenolic compounds in the susceptibility of bananas to crown rot. A review

Notes de la rédaction
Received on June 22, 2011; accepted on March 6, 2012
Résumé
Implication des composés phénoliques dans la sensibilité des bananes à la pourriture de couronne (synthèse bibliographique). La pourriture de couronne des bananes provoquée par un complexe parasitaire fongique est l'un des principaux défauts de qualité des bananes d'exportation. Des variations de sensibilité importantes des bananes à la pourriture de couronne ont été observées dans différentes zones de production. Il a été suggéré que l'état physiologique du fruit à la récolte influence la réponse des bananes à l'attaque des micro-organismes pathogènes et module ainsi sa sensibilité à la pourriture de couronne. La sensibilité des bananes à la pourriture de couronne est influencée par plusieurs facteurs pré-récolte, bien que les mécanismes de défense sous-jacents n'aient pas été clairement identifiés. Récemment, une étude basée sur des analyses moléculaires a suggéré que les composés phénoliques pouvaient être potentiellement impliqués dans les variations de sensibilités des bananes aux pourritures de couronne. D’autres études antérieures avaient déjà suggéré l'implication de composés phénoliques dans les réactions de défense du bananier contre diverses attaques de pathogènes. Cette étude se propose de faire l'état de l'art sur les variations de sensibilité des bananes à la pourriture de couronne, et de faire le point sur les composés phénoliques et leur implication potentielle dans les mécanismes de défense du bananier.
Abstract
Crown rot of bananas, caused by a fungal parasitic complex, is one of the main quality defects of exported bananas. Major variations in the susceptibility of bananas to crown rot have been observed in different production zones. The physiological state of the banana fruit at harvest is said to influence its response to pathogenic attack and thus to modulate its susceptibility to crown rot. The susceptibility of bananas to this disease, however, appears to be influenced by many pre-harvest factors, although the underlying defense mechanisms have not been clearly identified. A recent report based on molecular analyses suggests that phenolic compounds might be involved in the different variations in the susceptibility of bananas to crown rot. Results of other earlier studies point to an involvement of phenolic compounds in the defensive reactions of banana plants against various pathogens. The present paper reviews the current state of knowledge on the variations in the susceptibility of bananas to crown rot and takes stock of what is known about phenolic compounds in relation to their potential involvement in the defense mechanisms of the banana plant.
Table des matières
1. Introduction
1Banana is cultivated in more than 120 countries over about 10 million hectares. It ranks first in global fruit production with just over 106 million tons being produced annually worldwide. Cropping systems around the world are diverse and production targets include subsistence, sale in local or national markets, and international export. Some bananas, such as plantains, can be cooked, while others are classified as “dessert” bananas. Dessert bananas for export belong exclusively to the Cavendish subgroup and are subjected to an important international trade (Loeillet, 2005). Banana is currently, in terms of volume, the first exported fruit and ranks second after citrus fruits in terms of value. Total world exports of banana in 2006 accounted for 16.8 million tons (FAOSTAT data, 2006). Thus the banana industry is of vital importance to banana-producing countries.
2Banana trees and fruits are susceptible to attack by several diseases, including two main post-harvest diseases, namely banana anthracnose and crown rot. In Cameroon, the main quality defect that affects exported banana is crown rot. This disease is caused by a broad unspecific and opportunistic fungal parasitic complex. Fruit contamination can occur within the field but it mostly occurs in the washing tanks at the packing station where processing favors the penetration of pathogens into the crown tissues. The banana crowns, which were healthy at harvest, will then develop a fungal infection after a few days of shipping. Upon arrival, the quality of the banana fruits does not allow them to have a good position in the European market, and this sometimes constitutes a blow to exports. The use of synthetic fungicides to control banana tree diseases and post-harvest diseases is systematic. However, this approach is not satisfactory and is increasingly being criticized for the amounts of chemicals used, which could be harmful to humans as well as to the environment. Consequently, several approaches have been developed in recent years to find alternatives to chemical control.
3Variations have been noticed in the development of crown rot disease partly related to environmental conditions during growth. The occurrence of crown rot therefore depends firstly, on the level of susceptibility of the banana fruit to being influenced physiologically by environmental conditions during growth and secondly, on the level of contamination. Occurrence of crown rot also depends on the amount of defensive compounds (phenolics) accumulated in fruit tissues from flowering to harvest to fight pathogenic infections. Indeed, plant defense strategies involve the combination of several structural and biochemical mechanisms, including accumulated phenolic compounds and lignin-like polymers. However, with the current state of knowledge, it is extremely difficult to determine the mechanism responsible for the variability in the susceptibility of the fruit to crown rot.
4Up-to-date information will be presented below both on pre-harvest factors that influence the susceptibility of bananas to crown rot (especially biotic and abiotic factors) and on the involvement of phenolic compounds in plant defense mechanisms in relation to environmental conditions that support the plant’s growth.
2. Banana crown rot
5Exported bananas are subjected to various phytosanitary constraints throughout the production chain and these alter fruit marketability. Crown rot disease in particular causes major damage and economic losses in most banana-producing countries (Krauss et al., 2000). Losses of more than 86% have been reported for non-chemically-treated bananas exported from the Philippines (Alvindia et al., 2000).
2.1. Crown rot disease
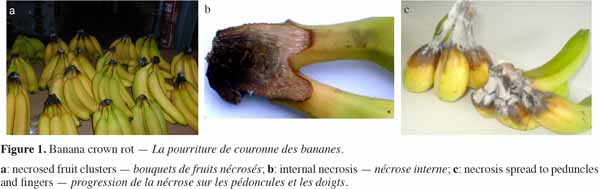
6Crown rot is a disease of the tissue uniting the peduncles. It occurs when banana hands are detached from their central axis, called the stalk. Banana hands are cut in clusters of 5-7 fruits, causing the exposure of wounded crown tissues to fungal infection (Muirhead et al., 2000). Fungal colonization of the crown starts afterwards, and the degeneration of the fruits does not require highly specialized parasites. Indeed, crown rot is a complex infection, which results from the activity of several microorganisms, of which Colletotrichum musae is the most pathogenic species (Finlay et al., 1993; Krauss et al., 2000; Muirhead et al., 2000; Lassois et al., 2010a). The infection is, however, not visible when the bananas are placed in commercial boxes and the first symptoms do not appear until after a few days of shipping. The disease evolves quickly during ripening. The rot is initially superficial and later progresses into the tissues. In some cases, it can invade the peduncles and later the fruits (Figure 1). The main effects on fruit quality are seen in rottening and in the induction of early ripening during shipping because of the ethylene that is released, either by stressed tissues and necrosed fruits or by fungal mycelium. When crown rot is frequently observed in bananas of a specific origin, production of bananas from this origin may be excluded from the market.
7Crown rot disease is currently controlled by the systematic use of post-harvest fungicidal treatments. This approach, however, has several drawbacks:
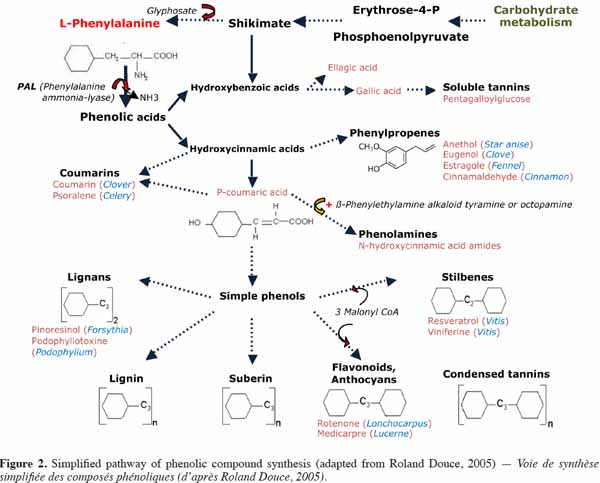
8– the effectiveness of fungicides depends on the time of year that they are applied and on the production area;
9– the frequent appearance of fungicide-resistant strains prevents complete disease control;
10– the fungicidal slurries rejected around packing stations after post-harvest treatment of banana crowns may cause environmental pollution.
11In addition, these post-harvest chemical sanitation treatments may have wider-reaching effects, with chemical exposure affecting workers at the packing stations, and even consumers. Evidence of the possible effect on consumers can be seen in the fungicidal residues regularly detected on banana peel and also in the pulp. These drawbacks have led to research focusing on alternative control methods.
2.2. Susceptibility of bananas to crown rot
12Variations in disease incidence. Geographical and seasonal variations have been observed in the incidence of crown rot disease (Lukezic et al., 1967; Shillingford, 1978; Krauss et al., 2000; Lassois et al., 2008). In a study conducted in Honduras, Lukezic et al. (1967) showed that the incidence of the disease varied in the course of the year, being higher in summer and lower during the colder period. They also found these variations to be unrelated to variations in the parasitic complex isolated from banana crowns. Similarly, in Jamaica, high levels of disease incidence were found to correlate with periods of high temperature (Shillingford, 1978). It has been hypothesized that such spatiotemporal fluctuations in disease incidence could reflect the variations in the quality potential of the fruit, acquired in the field (Lassois et al., 2010a). This quality potential would include two components, which would themselves depend on the agrotechnical and pedoclimatic factors prevailing during the growth phase of the banana plant. The first of these components is parasitic and the second is physiological. The parasitic component reflects the level of crown contamination by the fungal complex and the pathogenicity of this complex. The physiological component, for its part, reflects the fruit’s response to pathogenic attack, which is related to the physiological state of the fruit at harvest, and this state modulates the fruit’s susceptibility to crown rot.
13Variations in the level of fruit susceptibility. Recently, it has been shown that several pre-harvest factors influence the susceptibility of bananas to crown rot. These factors are: geographical and seasonal variations; bunch age; source-sink ratio; and biotic stress factors.
14Geographical variation. A multilocal study carried out in Cameroon over a year showed lesser crown rot development at higher altitude (500 m) than at lower altitude (80 m) (Ewané et al., submitted).
15Seasonal variation. In their study, Lassois et al. (2008) showed that the level of fruit susceptibility to crown rot disease was variable over a 10-week period in the same banana plot in Guadeloupe. The multilocal study carried out in Cameroon suggested that fruits harvested during the rainy season might be more susceptible to crown rot than those harvested during the dry season (Ewané et al., submitted).
16Bunch age and source-sink (So-Si) ratio. A within-bunch gradient of fruit is susceptible to crown rot. The first hands to initiate are more susceptible than those that initiate last (Lassois et al., 2010a). On the other hand, the So-Si ratio of the banana tree during its growth phase also has a significant influence on fruit susceptibility (hands being regarded as the sinks and leaves as the sources). When the total sink is decreased by the removal of banana hands from the bunch, the fruit’s susceptibility to crown rot also decreases. Moreover, a linear relationship between fruit age and fruit susceptibility to crown rot has also been demonstrated in Guadeloupe, the oldest fruits being the most susceptible (Forret, 2008).
17Biotic stress factors. The incidence of Mycosphaerella leaf spot disease also has an influence on crown rot disease, which is generated by host-pathogen interaction (Ewané et al., submitted).
18Among the great diversity of reactions and products involved in plant defense responses, phenolic compounds have been repeatedly proposed as potential participants in banana tree defense mechanisms (Beverragi et al., 1995; El Hadrami, 1997; Valette et al., 1998; Collingborn et al., 2000; Kanazawa et al., 2000; Someya et al., 2002; de Ascensao et al., 2003; Wuyts et al., 2007; Kavino et al., 2009). A recent report suggested that these compounds might contribute to determining the strength of the response of bananas to crown rot (Lassois et al., 2011).
3. Phenolic compounds
19Phenolic compounds are the most studied secondary metabolites because of their considerable involvement in plant-environment interactions. They are molecules belonging to very diverse chemical families having in common an aromatic ring bearing at least one phenol hydroxyl substituent. Some phenolic compounds have several hydroxyl group substituents, which can undergo esterification, methylation, etherification or glycosylation reactions (Raven et al., 2003; Macheix et al., 2005; Lattanzio et al., 2006). The molecular weight of phenolic compounds is variable, being lower in simple compounds, higher in those with complex structures, and higher still in polymerized tannins.
3.1. Classes of phenolic compounds
20Phenolic compounds are classified according to:
21– the nature and complexity of the carbonaceous skeleton;
22– the degree of skeletal modification (degree of oxidation, hydroxylation, methylation, etc.);
23– the link between the base unit and other molecules such as carbohydrates, lipids, proteins, or the link to other secondary metabolites, possibly polyphenols (Macheix et al., 2005).
24The phenolic compounds of plants include the simplest forms (hydroxybenzoic and hydroxycinnamic acids), condensed forms (tannins), and forms related to non-phenolic macromolecules (certain glucidic components of the pecto-cellulosic wall, cutin and suberin). Only two groups of phenolic compounds are mainly related to the plant walls: firstly, low-molecular-weight hydroxycinnamic acids related to various cell wall compounds, and secondly, lignins, which are polymers of monolignol units bound by oxidative coupling (Figure 2).
25Simple phenols (C6). These are compounds with one (monophenol-like catechin) or several phenolic groups (di, tri- and oligophenols): phenol, benzoquinone, pyrogallol, pyrocathecol, etc. (Lattanzio et al., 2006).
26Phenolic acids (C6-C1 or C6-C3). These are benzoic or hydroxybenzoic acids (gallic acid, ellagic acid), and cinnamic or hydroxycinnamic acids such as caffeic, coumaric, ferulic, and chlorogenic acid (Manach et al., 2004; Macheix et al., 2005; Lattanzio et al., 2006). A group of small phenolic molecules is derived from the subclass of hydroxycinnamic acids and is called phenylpropenes.
27Flavonoids (C6-C3-C6). These are present in plant vacuoles, where they are sometimes water-soluble or sometimes act as pigments (Raven et al., 2003). Flavonoids are the most abundant phenolic compounds in nature and are classified according to the degree of oxidation and unsaturation of their heterocyclic ring (Scalbert et al., 2000). Two classes of flavonoids can be distinguished: 4-oxoflavonoids and anthocyanidins (Manach et al., 2004).
28Lignins (C6-C3)n. These are extremely complex phenolic polymers. Of the biopolymers, lignins rank second in abundance after cellulose. The synthesis of these compounds results from a three-dimensional polymerization of three basic phenolic molecules (called monolignols): coumarylic, coniferylic and sinapylic alcohol, corresponding respectively to p-coumaric, ferulic and sinapic acid (Macheix et al., 2005). The complexity of lignins results from the potential association of these units via various chemical bonds, in a manner that is neither ordered nor repetitive, so as to generate an amorphous, hydrophobic polymer.
29Tannins (C6-C3-C6)n. These are found in several forms with different types of chemical reactivity and composition: water-soluble tannins, condensed tannins, catechic tannins and proanthocyanidins (Macheix et al., 2005). Proanthocyanidins have a high molecular weight and are a group of condensed (chain dimers or oligomers) flavan-3-ols often related to cell walls.
30The variability of phenolic compound classes in plants is far more complex than presented above. This is only intended to familiarize the reader with the classes of compounds relevant to the research topic.
3.2. Biosynthesis of phenolic compounds
31The biosynthesis pathway of phenolic compounds is well characterized (Figure 2). Phenolic compounds are formed via the well-known shikimate pathway from simple sugars resulting from primary metabolism. Synthesis of phenylalanine begins with carbohydrates transforming into erythrose-4-phosphate (pentose phosphate pathway) and phosphoenolpyruvate (glycolysis), which enter into the shikimic acid metabolic pathway. Phenylalanine deamination by phenylalanine ammonia-lyase (PAL) is the first crucial stage in the biosynthesis of the large majority of phenolic compounds (Macheix et al., 2005). In addition to its involvement in the production of phenolic compounds, phenylalanine oxidation also leads to the formation of tyrosine, the precursor of catecholamines (dopamine and derivatives) (Kulma et al., 2007), which are abundant compounds in the banana tree and fruit (Muirhead et al., 1984; Kanazawa et al., 2000; Wuyts et al., 2007).
32In plants, the phenolic composition varies considerably both qualitatively and quantitatively between species and between individuals of the same species. Some phenolic compounds are ubiquitous in plants (e.g. hydroxycinnamic acids), some are common (e.g. anthocyanins) and others are specific to certain families or species (e.g. isoflavonoids and stilbenes). Besides plant variety, a wide range of biotic and abiotic stresses (physical, chemical and biological factors) both external and endogenous can influence the level of plant phenolic content both before and after harvest. Changes in the level of plant phenolic content are effected through modulation of the phenolics metabolism: light (visible and UV), temperature, osmotic potential, plant nutrition, growth regulators, biotic elicitors, the fruit maturation state at harvest, the photoperiod, moisture, processing, and storage (Manach et al., 2004; Kalt, 2005; Macheix et al., 2005; Lattanzio et al., 2006; Ksouri et al., 2008; Xu et al., 2008; He et al., 2010; Treutter, 2010). This leads to an alteration of a very broad palette of functions, including the release or synthesis of stress metabolites involving polyphenols. For example, environmental stresses can affect photosynthesis and the assimilation of carbon required for growth, development and defense, leading to an influence on the final phenolics concentration in plant tissues. In the following sections, the influence of biotic and abiotic factors on phenolics synthesis is fully discussed.
33Influence of biotic factors. Plants face daily parasitic attacks by pathogenic microorganisms, herbivores and insects. Parasites have developed different strategies for colonizing plants, since they need regulation mechanisms to effectively adapt to changes in their environment. Development of a pathogen within a host plant occurs only under certain conditions. These conditions relate to the environmental factors influencing the host plant’s physiological state, the parasite’s capacity to attack and colonize its host, and the host’s capacity to establish mechanisms capable of stopping colonization by the parasite. Plants have, however, developed preformed and induced defense mechanisms effective against pathogenic infections.
34Following penetration by pathogens of the physical and chemical constitutive barriers of a susceptible or a resistant plant, induced structural and chemical barriers are activated in order to prevent the pathogen’s progression. This results in a multitude of defense responses. For instance, the defense response of the tomato tissue is seen in an increase in soluble phenol (Zhu et al., 2004), and/or the deposit of lignin and lignin polymers (Mandal et al., 2007; Panina et al., 2007). When a plant cell encounters a pathogen, this also provokes other reactions such as the hypersensitive response (HR), which may confine the pathogen at the penetration point (Lepoivre, 2003; Lattanzio et al., 2006; Van Loon et al., 2006; Ferreira et al., 2007). However, phenolic compounds have different rates of accumulation depending on whether plant-pathogen interaction is compatible or incompatible.
35The HR triggers a general resistance mechanism, rendering uninfected parts of the plant less sensitive to further attack by pathogens (Lattanzio et al., 2006). Resistant reactions involving phenolic compounds are induced in the adjacent cells surrounding the infected zone, forming a localized acquired resistance (LAR). In distal tissue cells, a systemic acquired resistance (SAR) is established (Lepoivre, 2003; Macheix et al., 2005; Lattanzio et al., 2006; Van Loon et al., 2006; Ferreira et al., 2007). Stilbene phytoalexines (resveratrol), for example, are accumulated in grapewine berries following fungal infection by Botrytis cinerea, but also in healthy grapes on the same cluster or near infected grape clusters (Adrian et al., 2000). In the case of the banana tree, the presence of Mycosphaerella leaf spot diseases during growth influences the susceptibility of bananas to post-harvest diseases (Ewané et al., submitted), but the possible involvement of phenolic compounds has not been investigated.
36Influence of abiotic factors. Abiotic factors – e.g. pedoclimatic, agronomic and agrotechnical − influence the phenolic biosynthetic process in plant tissue cells before and after harvest. The geographical location of the production zone can influence the biosynthesis of polyphenols in plant tissues. Tropical and high altitude plants contain higher proportions of flavonoids than do temperate plants (Lattanzio et al., 2006), probably because of differences in climatic regimes.
37Seasonal variability in plant phenolic content may be due to the effects of climatic factors such as solar radiation, temperature, rainfall (water availability) and the hydrothermal coefficient between seasons. Spatiotemporal variations have been found to influence the susceptibility of bananas to crown rot, but further investigations are needed to provide evidence for the influence of these variations on the fruit’s phenolic content. A wide array of abiotic stimuli, such as salinity and drought, are capable of triggering changes in the plant’s metabolism and these changes enhance the production of plant secondary products (Khan et al., 2011).
38High levels of solar radiation coupled with high temperatures lead in some cases to modifications in the accumulation of polyphenols in the plant. The resulting modifications make it difficult to determine whether physiological changes related to phenolic synthesis in the plant are caused by the effects of either solar radiation or high temperature. The literature has reported that temperature can be positively or negatively correlated to the accumulation of polyphenols during growth or processing for several plant species (Kalt, 2005; He et al., 2010; Treutter, 2010). A prominent example of the effect of temperature and light can be seen in the significantly high level of phenolics found in the seed and skin of winter berries compared to summer berries (Xu et al., 2011).
39Some information on the effect of light exposure, UV and solar radiation on PAL and phenolics synthesis has been reported in the literature for several plant species. The influence of other factors has also been cited, such as soil fertility, fertilization mode (nutrient supplies), irrigation (water availability), rootstocks, elevated atmospheric CO2, and pre-harvest treatment (Kalt, 2005; Lattanzio et al., 2006; He et al., 2010; Treutter, 2010; Pombo et al., 2011). Hence, it is not only the energy provided for carbon assimilation (carbon resources for biosynthesis) but also the quality, namely UV fractions (light intensity), which stimulates the formation and accumulation of certain phenolic compounds in plants (Ghasemzadeh et al., 2010; Treutter, 2010). The influence of soil with high levels of aluminum chloride in grape leaves has been shown to stimulate the production of resveratrol (Adrian et al., 2000).
40Cultural practice is another factor that influences the phenolic content of plants and allows the control of plant growing conditions. Since carbohydrate availability is a prerequisite for phenylpropanoid accumulation (Treutter, 2010), the degree of ripeness or physiological age of the fruit at harvest (maturity) has a considerable effect on the concentration and proportions of the various polyphenols (Macheix et al., 1990; Macheix et al., 2005). Moreover, So-Si ratio modification has been shown to improve the chemical composition of Vitis vinifera cultivars through an impact on anthocyanin accumulation (Mota et al., 2010). So-Si ratio modification also influences the susceptibility of the banana fruit to crown rot (Lassois et al., 2010b). An evaluation of the level of phenolic compound synthesis has not yet been undertaken. Furthermore, cultural conditions such as organic or sustainable agriculture provide higher amounts of polyphenols in strawberries, blackberries, and corn than do conventional or hydroponic conditions without stress (Asami et al., 2003).
41Several studies have reported the influence of processing and storage on the synthesis of phenolic compounds or on degradation in fruits, vegetables and plants (Kalt, 2005; Lattanzio et al., 2006; He et al., 2010; Treutter, 2010). At the packing station, during the preparation of banana clusters, physical injury to the fruit through cutting of the crown tissues may lead to oxidative degradation of polyphenols. This oxidative degradation of polyphenols causes their transformation into brown pigments that are polymerized to different degrees or else it causes polyphenol synthesis in the banana tissues. In addition, certain water-soluble phenolics present in the wounded crown may leach from fruit tissues into washing tanks. Finally, the number of phenolic compounds known to be synthesized and stored by higher plants is in the thousands and rising; however, the functions of many phenolics remain unknown.
3.3. Localization and function of phenolic compounds in plants
42Biosynthesis of phenolic compounds occurs at various sites in plant cells, such as the chloroplasts, the cytoplasm and the endoplasmic reticulum membrane. Polyphenols (relatively hydrophilic) usually accumulate in the central vacuoles of guard cells, epidermal cells and the subepidermal cells of leaves and shoots. Some polyphenols are found covalently linked to the plant cell wall (lignin); others are found in waxes (related to lipidic structures) or on the external surfaces (cuticle) of plant organs (Lattanzio et al., 2006). The localization of a phenolic compound within a tissue reflects its physiological function or its participation in interactions of the plant with its environment (Macheix et al., 2005). For example, polyphenols with a role in signaling or defense are often stored at strategically important sites (Lattanzio et al., 2006).
43Phenolic compounds, both preformed and induced, play an essential role in the balance of the plant within its environment and its capacity to adapt to environmental changes (Macheix et al., 2005) through biochemical, physiological and molecular responses. Phenolic compounds intervene in major physiological mechanisms of the plant, such as growth, reproduction, pigmentation, rhizogenesis, vitrification and resistance to pathogens, by performing many functions (Cheynier, 2005; Lattanzio et al., 2006). Polyphenols play a key role as the major red, blue, and purple pigments of plants, as antioxidants and metal chelators, and as agents acting both above and below ground in signaling between the plant and other organisms and as UV light screens (Cheynier, 2005; Lattanzio et al., 2006). Polyphenols have multiple effects on tissue maturation processes and on the sensory qualities of plant-derived food products, including astringency, bitterness and aroma. The compounds are also responsible for the browning caused by wounds to fruit or by conservation accidents and notably for the effect of low temperatures on pineapple and banana. In banana, dopamine has been identified as a substrate for polyphenol oxidase, resulting in the browning reaction, and the chemical is also responsible for tissue discoloration (Kanazawa et al., 2000; Someya et al., 2002; Maneenuam et al., 2007). As polyphenols play a role in regulating plant-environment interactions, some researchers attribute an ecological function to these compounds. Polyphenols are, for instance, reported to modulate litter decomposition processes and the mineralization rate in plants (Macheix et al., 2005; Lattanzio et al., 2006). Plants thus contain several classes of polyphenols with different localizations and roles.
3.4. Involvement of phenolic compounds in plant defense mechanisms
44Phenolic compounds involved in plant defense are either preformed (constitutive) or synthesized de novo (postinfectional). Preexisting phenols are antibiotic (antifungal) compounds such as simple phenols, phenolic acids, flavonols, and dihydrochalcones. These preexisting phenols are called phytoanticipins in order to distinguish them from phytoalexins, which are synthesized from remote precursors in response to pathogenic attack (Lattanzio et al., 2006). Phenolic compounds synthesized de novo accumulate in response to plant infection by a pathogen. This defensive response involves the rapid increase of specific phenolic compounds at the infected site, particularly phytoalexins, which can inhibit a broad range of microorganisms (Lepoivre, 2003; Macheix et al., 2005; Lattanzio et al., 2006). The result is the development of plant resistance to disease. An example of the involvement of polyphenols in plant defense is their action in the programmed cell death of one part of the plant. The rate at which programmed cell death occurs depends on whether the host-pathogen interaction is compatible or incompatible. During the establishment of a pathogen in host tissues, there is an increase in the activity of specific enzymes such as PAL, peroxidase and polyphenol oxidase. These enzymes, which consume oxygen and produce fungitoxic quinones, make the medium unfavorable for the further development of pathogens (Lattanzio et al., 2006). PAL is the key enzyme involved in phenolic compound metabolism through the phenylpropanoid pathway (Dixon et al., 2002). Peroxidase catalyzes the condensation of phenol into lignin and is also involved in phenol metabolism (Passardi et al., 2004). Polyphenol oxidase oxidizes constitutive plant phenols into quinones, which have bactericidal and fungicidal properties, and is also involved in the oxidative detoxification of pathogen phytotoxins (Macheix et al., 1990; Yoruk et al., 2003).
45Another example of a phenolic compound that contributes to the plant’s defense mechanisms is lignin. Lignin is a phenolic polymer, which plays a fundamental role in solute conductance, mechanical support and disease resistance. In response to abiotic stress, to wounding or to pathogenic infection, the deposition of lignins, lignin polymers and other phenolic substances related to the cell wall are observed. This contributes to both a thickening of the cell wall (conferring greater rigidity and mechanical resistance) and to an increase in cell hydrophobicity. Lignin thus acts as a physical barrier against pathogenic invasion. In addition, lignin deposits reduce the diffusion of enzymes and toxins that the pathogen releases in order to facilitate host tissue colonization. Lignin also deprives the pathogen of the plant water and nutrients necessary to its proliferation (Macheix et al., 2005; Lattanzio et al., 2006).
3.5. Involvement of phenolic compounds in banana tree defense
46Since phenolic compounds are potential targets for the improvement of plant resistance against pathogenic attack, it is possible that they are involved in the susceptibility of bananas to crown rot disease and a better understanding of their involvement in banana tree defense is required. The phenolic composition and the role of several banana tree tissues (leaves, fruits, roots) have been described in the literature review, but their potential role in the variation of susceptibility of banana crowns to disease is not yet known (Table 1).
47Preformed defense mechanisms. The leaf surface wax, cuticle, suberin and cell wall are the first preformed physical barriers in banana. These preformed physical barriers are also involved in banana tree defense mechanisms, and contain phenolic compounds (lignin, cross-linking hydroxycinnamic acids) in their structures. The cuticle forms an envelope that protects the aerial parts of plants from the majority of attacks by pathogenic fungi (Pollard et al., 2008). Suberin, localized in the underground parts of the plants such as the bark and scar tissues, contains high quantities of ω-dicarboxylic acid, hydroxycinnamic acids (mainly ferulate) and fatty alcohols (Pollard et al., 2008; Schreiber, 2010). The cuticle and suberin control the diffusion of gases, of water, and of solutes in and out of the plant. They also play a protective role towards biotic and abiotic stress (Pollard et al., 2008; Schreiber, 2010). The pecto-cellulosic wall plays the role of “skeleton” while maintaining a certain plasticity/elasticity in the plant, allowing growth and cellular divisions (Macheix et al., 2005), but also being involved in physiological resistance.
48The preformed chemical barriers of plants are a plethora of peptides, proteins and secondary metabolites (phenolics, sulfur compounds, etc.), which act as antimicrobial compounds during defense responses against microorganisms (Lattanzio et al., 2006). Some of these compounds are present in their biologically active form, and are therefore directly toxic; others are present in the form of inactive precursors (combined forms). In studies comparing varieties of banana susceptible to black leaf streak disease with partially resistant varieties, certain mesophyll cells specialized in the storage of phenolic compounds (proanthocyanidins) were found to be lower in number in the susceptible varieties than in the partially resistant ones (Beveraggi et al., 1995; El Hadrami, 1997).
49Induced defense mechanisms. After infection by different banana pathogens, a rapid increase in both phenolic compounds and phenylpropanoid pathway enzymes has been observed in banana tree tissues. Induced structural barriers are activated following the presence of pathogens in susceptible or resistant plants, and this results in a multitude of defense responses. When a plant recognizes a pathogenic invader, its defense mechanisms are induced, and chemical barriers are produced as a result. These chemical barriers are generally in the form of "de novo'' synthesized molecules and once produced, these molecules become accumulated.
50Several studies on the interaction between banana tree roots and their various pathogens have suggested an involvement of phenolic compounds in the defense responses of the banana tree. These studies focused on cell wall lignification, on histochemical detection of phenolic compounds and on the quantification of flavonoids, proanthocyanidins and hydroxycinnamic acids after infection of banana roots by phytophagous nematodes (Valette et al., 1998; Collingborn et al., 2000; Kanazawa et al., 2000; de Ascenceo et al., 2003; Wuyts et al., 2007). In resistant banana cultivars, a higher content of several phenolic compounds such as dopamine and cyanidin-related compounds, lignin, and ferulic acid (esters and hydrolyzation products of ferulic acid) was observed (Wuyts et al., 2007). Moreover, the role of these compounds in defense mechanisms such as physical barriers, toxic agent and protection against pathogenic attack was suggested.
51In other studies, several phytoalexins (phenylphenalenones, irenolone, and emenolone types) have been induced in green banana fruits after wounding or inoculation with Colletotrichum musae (Luis et al., 1993; Kamo et al., 1998; Kamo et al., 2001). Recently, in the interaction between Musa spp. and Fusarium oxysporum f. sp. cubense (Panama disease), resistant hybrid plantlets (leaves and roots) were found to contain higher concentrations of phenol and pathogenesis-related proteins and higher activity levels of PAL and oxidative enzymes (peroxidase, polyphenol oxidase, superoxide dismutase, catalase) than sensitive hybrid plantlets (Kavino et al., 2009). During penetration of host tissues, phenolic substances are released around the pathogen hyphae, causing considerable morphological changes such as cytoplasmic disorganization and loss of protoplasmic contents (Kavino et al., 2009) and thus forming a chemical barrier that blocks or at least slows down microorganism progression.
52As mentioned above, many phenolic compounds, particularly dopamine, have been identified in various banana tree tissues. Among a broad range of plants, bananas have the richest dopamine content: 100 μg·g-1 FW in Cavendish banana peel as compared with 7 μg·g-1 FW for potato, the second richest in this list of dopamine-rich plants (Kulma et al., 2007). This observation suggests that dopamine might play an important role in banana physiology. Dopamine results from the hydroxylation of tyrosine to 3,4-dihydroxyphenylalanine, and is responsible for the browning of the fruit peel and pulp. Products arising from the decomposition of banana dopamine seem to have a fungitoxic activity against C. musae (Mulvena et al., 1969; Muirhead et al., 1984). Moreover, a recent study on the molecular mechanisms involved in the variations in the susceptibility of banana fruit has highlighted overexpression of the dopamine-β-monooxygenase gene in bananas less susceptible to crown rot (Lassois et al., 2011).
4. Conclusion
53The occurrence of crown rot disease seems to correlate with the physiological state of the fruits at harvest. Plant polyphenols are secondary metabolites involved in the defense mechanisms of plants against fungal pathogens. These phenolic compounds are also involved in the defense responses of the banana tree, although their mechanism(s) of action and their antagonistic properties are not known. Thus, the possible involvement of phenolic compounds in variations found in the susceptibility of bananas to crown rot means that these compounds represent a potential target for developing alternatives to the chemical control of the disease. It is thus essential to measure pools of phenolic compounds, particularly dopamine, in fruits displaying differential susceptibility, so as to identify key “defense” compounds and to gain a better knowledge of the defense mechanisms of the banana crown tissues.
Bibliographie
Adrian M. et al., 2000. Stilbene content of mature Vitis vinifera berries in response to UV-C elicitation. J. Agric. Food Chem., 48(12), 6103-6105.
Alvindia D.G., Kobayashi T., Yaguchi Y. & Natsuaki K.T., 2000. Symptoms and the associated fungi of post-harvest diseases on non-chemical bananas imported from the Philippines. Jpn. J. Trop. Agric., 44, 87-93.
Asami D.K., Hong Y.J., Barrett D.M. & Mitchell A.E., 2003. Comparison of the total phenolic and ascorbic acid content of freeze-dried and air-dried marionberry, strawberry, and corn grown using conventional, organic, and sustainable agricultural practices. J. Agric. Food Chem., 51, 1237-1241.
Beveraggi A., Mourichon X. & Sallé G., 1995. Étude comparée des premières étapes de l’infection chez des bananiers sensibles et résistants infectés par Cercospora fijiensis, agent de la maladie des raies noires. Can. J. Bot., 73, 1328-1337.
Cheynier V., 2005. Polyphenols in foods are more complex than often thought. Am. J. Clin. Nutr., 81, 223-229.
Collingborn F.M.B., Gowen S.R. & Mueller-Harvey I., 2000. Investigations into the biochemical basis for nematodes resistance in roots of three Musa cultivars in response to Radopholus similis infection. J. Agric. Food Chem., 48, 5297-5301.
de Ascensao A.R.F.D.C. & Dubery I.A., 2003. Soluble and wall-bound polyphenols and phenolic polymers in Musa acuminata roots exposed to elicitors from Fusarium oxysporum f. sp. cubense. Phytochemistry, 63, 679-686.
Dixon R.A. et al., 2002. The phenylpropanoid pathway and plant defence – a genomics perspective. Mol. Plant Pathol., 3, 371-390.
Douce R., 2005. Les plantes supérieures : divines et/ou diaboliques. Conférences et débats sur les grands défis du 21e siècle. Paris : Institut de France, Académie des Sciences.
El Hadrami A., 1997. Proanthocyanidines constitutives des feuilles de bananiers et résistance partielle vis-à-vis de Mycosphaerella fijiensis, l’agent causal de la maladie des raies noires. Mémoire : Faculté universitaire des Sciences agronomiques de Gembloux (Belgique).
FAOSTAT, 2006. FAOSTAT data. http://faostat.fao.org, (13/12/2011).
Ferreira R.B. et al., 2007. The role of plant defence proteins in fungal pathogenesis. Mol. Plant Pathol., 8, 677-700.
Finlay A.R. & Brown A.E., 1993. The relative importance of Colletotrichum musae as a crown rot pathogen on Windward Island bananas. Plant Pathol., 42, 67-74.
Forret M., 2008. Étude de la variation de sensibilité des bananes d’exportation aux pourritures de couronne en fonction du stade de récolte. Mémoire : Faculté universitaire des Sciences agronomiques de Gembloux (Belgique).
Ghasemzadeh A., Jaafar H.Z.E. & Rahmat A., 2010. Synthesis of phenolics and flavonoids in ginger (Zingiber officinale Roscoe) and their effects on photosynthesis rate. Int. J. Mol. Sci., 11, 4539-4555.
He F. et al., 2010. Biosynthesis of anthocyanins and their regulation in colored grapes. Molecules, 15, 9057-9091.
Kalt W., 2005. Effects of production and processing factors on major fruit and vegetable antioxidants. J. Food Sci., 70(1), 11-19.
Kamo T. et al., 1998. Phenylphelanone-type phytoalexins from unripe bungulan banana fruit. Biosci. Biotechnol. Biochem., 62, 95-101.
Kamo T. et al., 2001. New phenylphenalenones from banana fruit. Tetrahedron, 57, 7649-7656.
Kanazawa K. & Sakakibara H., 2000. High content of dopamine, a strong antioxidant, in Cavendish banana. J. Agric. Food Chem., 48, 844-848.
Kavino M. et al., 2009. Biochemical markers as a useful tool for the early identification of Fusarium oxysporum f.sp. cubense, race 1 resistance banana clones. Arch. Phytopathol. Plant Prot., 42(11), 1069-1078.
Khan T.A., Mazid M. & Mohammad F., 2011. Status of secondary plant products under abiotic stress: an overview. J. Stress Physiol. Biochem., 7(2), 75-98.
Krauss U. & Johanson A., 2000. Recent advances in the control of crown rot of banana in the Windward Islands. Crop Prot., 19, 151-160.
Ksouri R. et al., 2008. Influence of biological, environmental and technical factors on phenolic content and antioxidant activities of Tunisian halophytes. C.R. Biol., 331(11), 865-873.
Kulma A. & Szopa J., 2007. Catecholamines are active compounds in plants. Plant Sci., 172, 433-440.
Lassois L., de Lapeyre de Bellaire L. & Jijakli M.H., 2008. Biological control of crown rot of bananas with Pichia anomala strain K and Candida oleophila strain O. Biol. Control, 45, 410-418.
Lassois L. et al., 2010a. Hand position on the bunch and source-sink ratio influence the banana fruit susceptibility to crown rot disease. Ann. Appl. Biol., 156(2), 221-229.
Lassois L., Jijakli M.H., Chillet M. & de Lapeyre de Bellaire L., 2010b. Crown rot of bananas: pre-harvest factors involved in post-harvest disease development and integrated control methods. Plant Disease, 94(6), 648-658.
Lassois L. et al., 2011. Identification of genes involved in the response of banana to crown rot disease. Mol. Plant-Microbe Interact., 24, 143-153.
Lattanzio V., Lattanzio V.M.T. & Cardinali A., 2006. Role of polyphenols in the resistance mechanisms of plants against fungal pathogens and insects. In: Imperato F., ed. Phytochemistry: advances in research. Trivandrum, Kerala, India: Research Signpost, 23-67.
Lepoivre P., 2003. Phytopathologie : bases moléculaires et biologiques des pathosystèmes et fondements des stratégies de lutte. Bruxelles : Éditions De Boeck Université.
Loeillet D., 2005. Le commerce international de la banane : entre évolution et révolution. Fruitrop, 129, 2-19.
Luis J.G. et al., 1993. Irenolone and emenolone: two new types of phytoalexin from Musa paradisica. J. Org. Chem., 58, 4306-4308.
Lukezic F.L., Kaiser W.J. & Martinez M.M., 1967. The incidence of crown rot of boxed bananas in relation to microbial populations of the crown tissue. Can. J. Bot., 45, 413-421.
Macheix J., Fleuriet A. & Billot J., 1990. Phenolic compounds in fruit processing. In: Macheix J., Fleuriet A. & Billot J., eds. Fruit phenolics. Boca Raton, FL, USA: CRC Press, 239-312.
Macheix J.-J., Fleuriet A. & Jay-Allemand C., 2005. Les composés phénoliques des végétaux : un exemple de métabolites secondaires d'importance économique. Lausanne, Suisse : Presses Polytechniques et Universitaires Romandes.
Manach C. et al., 2004. Polyphenols: food sources and bioavailability. Am. J. Clin. Nutr., 79, 727-747.
Mandal S. & Mitra A., 2007. Reinforcement of cell in roots of Lycopersicon esculentum through induction of phenolics and lignin by elicitors. Physiol. Mol. Plant Pathol., 71, 201-209.
Maneenuam T., Ketsa S. & van Doorn W.G., 2007. High oxygen levels promote peel spotting in banana fruit. Postharvest Biol. Technol., 43, 128-132.
Mendez C.d.M.V. et al., 2003. Content of free phenolic compounds in bananas from Tenerife (Canary Islands) and Ecuador. Eur. Food Res. Technol., 217, 287-290.
Mendoza E.M.T. et al., 1992. Les polyphénols des bananes à cuire. Changements pendant le mûrissage et la cuisson et relations avec l'astringence. Philippines J. Crop Sci., 17(3), 155-161.
Mota R.V. et al., 2010. Biochemical and agronomical responses of grapevines to alteration of source-sink ratio by cluster thinning and shoot trimming. Bragantia Campinas, 69(1), 17-25.
Muirhead I.F. & Deverall B.J., 1984. Evaluation of 3,4-dihydroxybenzaldehyde, dopamine and its oxidation products as inhibitors of Colletotrichum musae (Berk. & Curt.) Arx. in green banana fruits. Aust. J. Bot., 32, 375-382.
Muirhead I.F. & Jones D.R., 2000. Fungal diseases of banana fruit. Post-harvest diseases. In: Jones D.R., ed. Diseases of banana, abaca and ensete. Wallingford, UK: CABI.
Mulvena D., Edwin C.W. & Zerner B., 1969. 4-dihydroxybenzaldehyde, a fungistatic substance from green Cavendish bananas. Phytochemistry, 8, 393-395.
Panina Y., Frave D.R., Baker C.J. & Shcherbakova L.A., 2007. Biocontrol and plant pathogenic Fusarium oxysporum-induced changes in phenolic compounds in tomato leaves and roots. J. Phytopathol., 155, 475-481.
Passardi F., Penel C. & Dunand C., 2004. Performing the paradoxical: how plant peroxidases modify the cell wall. Trends Plant Sci., 9, 534-540.
Pollard M., Beisson F., Li Y. & Ohlrogge J.B., 2008. Building lipid barriers: biosynthesis of cutin and suberin. Trends Plant Sci., 13, 236-246.
Pombo M.A., Rosli H.G., Martínez G.A. & Civello P.M., 2011. UV-C treatment affects the expression and activity of defence genes in strawberry fruit (Fragaria×ananassa, Duch.). Postharvest Biol. Technol., 59, 94-102.
Raven P.H., Evert R.F., Eichhorn S.E. & Bouharmont J., 2003. Biologie végétale. Bruxelles : Éditions De Boeck Université.
Scalbert A. & Williamson G., 2000. Dietary intake and bioavailability of polyphenols. J. Nutr., 130, 2070S-2085S.
Schreiber L., 2010. Transport barriers made of cutin, suberin and associated waxes. Trends Plant Sci., 15, 546-553.
Shillingford C.A., 1978. Climatic factors affecting post-harvest decay of Jamaican bananas. J. Agric. Univ. Puerto Rico, 63(1), 45-49.
Someya S., Yoshiki Y. & Okubo K., 2002. Antioxydant compounds from bananas (Musa cavendish). Food Chem., 79, 351-354.
Treutter D., 2010. Managing phenol contents in crop plants by phytochemical farming and breeding-visions and constraints. Int. J. Mol. Sci., 11, 807-857.
Valette C. et al., 1998. Histochemical and cytochemical investigations of phenols in roots of banana infected by burrowing nematode Radopholus similis. Phytopathology, 88, 1141-1148.
Van Loon L.C., Rep M. & Pieterse C.M.J., 2006. Significance of inducible defence-related proteins in infected plants. Annu. Rev. Phytopathol., 44, 135-162.
Wade N.L., Tan S.C. & Kavanagh E.E., 1993. White light prevent increased catechin synthesis by ultraviolet irradiation in banana fruits. J. Hortic. Sci., 68, 637-644.
Wuyts N. et al., 2007. Potential physical and chemical barriers to infection by the burrowing nematode Radopholus similis in roots of susceptible and resistant banana (Musa spp.). Plant Pathol., 56(5), 878-890.
Xu C. et al., 2011. Influence of growing season on phenolic compounds and antioxidant properties of grape berries from vines grown in subtropical climate. J. Agric. Food Chem., 59(4), 1078-1086.
Xu F. et al., 2008. Molecular cloning, characterization and expression of phenylalanine ammonia-lyase gene from Ginkgo biloba. Afr. J. Biotechnol., 7(6), 721-729.
Yoruk R. & Marshal M.R., 2003. Physicochemical properties and function of plant polyphenol oxidase: a review. J. Food Biochem., 27, 361-422.
Zhu H.H. & Yao Q., 2004. Localized and systemic increase of phenols in tomato roots induced by Glomus versiforme inhibits Ralstonia solanacearum. J. Phytopathol., 152, 537-542.
Pour citer cet article
A propos de : Cécile Annie Ewané
Univ. Liege - Gembloux Agro-Bio Tech. Plant Pathology Unit. Avenue Maréchal Juin, 13. B-5030 Gembloux (Belgium). E-mail: cewanea@yahoo.fr — CARBAP. African Research Center on Bananas and Plantains. Plant Pathology Unit. P.O. Box 832. CAM-Douala (Cameroon) — University of Yaounde I. Faculty of Sciences. Department of Biochemistry. P.O. Box 812. CAM-Yaounde (Cameroon).
A propos de : Philippe Lepoivre
Univ. Liege - Gembloux Agro-Bio Tech. Plant Pathology Unit. Avenue Maréchal Juin, 13. B-5030 Gembloux (Belgium).
A propos de : Luc de Lapeyre de Bellaire
CIRAD. Research Unit Banana, Plantain and Pineapple Cropping Systems. TA B-26/PS4 (Bur. 108). Boulevard de la Lironde. F-34398 Montpellier Cedex 5 (France).
A propos de : Ludivine Lassois
Univ. Liege - Gembloux Agro-Bio Tech. Plant Pathology Unit. Avenue Maréchal Juin, 13. B-5030 Gembloux (Belgium).





