- Accueil
- Volume 11 (2007)
- numéro 2
- Dormancy in sweet cherry (Prunus avium L.) under Mediterranean climatic conditions
Visualisation(s): 5763 (31 ULiège)
Téléchargement(s): 256 (2 ULiège)
Dormancy in sweet cherry (Prunus avium L.) under Mediterranean climatic conditions

Notes de la rédaction
Received on 24 August 2004, accepted on 20 February 2006.
Résumé
La dormance chez le cerisier Prunus avium L. en climat mediterranéen. On admet qu'une cause importante de l'inadaptation des espèces fruitières d'origine tempérée en zone chaude est la non satisfaction de leurs besoins de froid. Toutefois, pour une espèce comme le cerisier, les caractéristiques de la dormance et de son évolution sous climat chaud sont encore mal connues. Nous avons contribué à leur connaissance par un suivi de la dormance des bourgeons chez un certain nombre de variétés de cerisier doux (Prunus avium L.) cultivées à Meknès (Maroc) en utilisant l'approche biologique du test « Boutures de nœuds ». Les variations du temps moyen de débourrement (TMD) sont interprétées en termes d’évolution des capacités de croissance des bourgeons. Le manque de froid affecte profondément le développement phénologique des bourgeons floraux. L’évolution de la dormance a montré l'existence de trois périodes d’inertie variable : la première et la troisième période, d’inertie et de durée similaire, alors que la seconde période d’inertie importante (endo-dormance). Les influences inhibitrices corrélatives des feuilles continuent à s’exercer sur les bourgeons jusqu’à l'arrivée du froid continu. La tardiveté de la diminution de l'inertie traduit une levée de dormance incomplète qui se manifeste, au niveau du verger, par un débourrement déficient et étalé des bourgeons. L’examen histologique de bourgeons floraux a montré que la connexion vasculaire est établie à l’anthèse. Cependant les fleurs ont montré des anomalies relatives à une faible production du pollen et à une malformation des pistils qui limitent les possibilités de pollinisation.
Abstract
It is admitted that the lack of winter chilling is a limiting factor for the cultivation of temperate fruit trees in warm climates. Nevertheless, the characteristics of dormancy in sweet cherry under such conditions are still not fully understood. Therefore, and in order to contribute to the elucidation of these mechanisms, the objective of this work is to evaluate the dormancy of sweet cherry (Prunus avium L.) under the Mediterranean conditions of Meknes region (Morocco). Evaluation of dormancy behaviour of four varieties was made on the basis of a biological test, known as the « single node cuttings », and histological dissections of flower buds at the time of bud break. The variations of mean time to bud break (MTB) were interpreted in terms of the evolution of growth capacity of the buds. The lack of cold affected tree phenological development. The evolution of dormancy in cherry buds showed three phases with variable levels of inertia: the first and third phases being of similar intensity and duration while the second phase was of a high level of inertia (endo-dormancy). The correlative inhibitions of the leaves imposed on the buds continued up to the arrival of continuous low temperatures. The delay in the decrease of inertia reveals insufficient chilling accumulation causing incomplete release from dormancy and low bud break percentage in the orchard. Histological observations showed that vascular connection of flower buds was established during bud break stage. However, flowers showed certain abnormalities, ie low pollen production and malformations of pistils, which limit the possibilities of pollination.
Table des matières
1. Introduction
1Fruit trees, like other woody perennials, are characterized by dormancy period. In contrast to endo-dormancy which concerns the bud itself, correlative inhibition results from the action of one or many other organs (stem, leaves, roots, other bud) (Crabbé, 1987). The release from dormancy requires the accumulation of chilling during the winter period (Crabbé, 1994). The development of correlative inhibitions during the summer period leads to the suspension of bud growth and the onset of dormancy. During fall and winter this dormancy is overcome under the effects of low temperatures. In the absence of adequate winter chilling, dormancy is prolonged, flower buds abscise and yield is lower. Increased chilling caused an increase in the number of branches and reduced the time to bud break. Small and deformed blossoms result also from inadequate chilling (Mahmood et al., 2000a).
2In Morocco, cherry was traditionally grown at higher elevation (> 1000 m) where the winters provide enough chilling to respond to the requirement of the species. However, and for economical raisons, the crop has been extended to lower elevations where the climatic conditions are not adequate for sweet cherry to produce. Thus, several orchards were established with standard varieties such as « Bigarreau Burlat », « B. Moreau » and « B. Napoleon ». Unfortunately, these varieties, because of the lack of winter chilling in these areas, showed symptoms of prolonged dormancy resulting in a vegetative growth which continues late in the season and a delayed bud break (Oukabli et al., 2002). The bud break is also sporadic and the vegetative growth is heterogeneous as certain buds (usually the apical ones) start to grow but impose on those located beneath a strong correlative inhibition. The latter prevents new bud to break or delays their growth if they are at an advanced stage. Thus, only a low proportion of buds start to grow. The dominant role of the terminal bud has been established in the control of growth and the strengthening of the apical dominance (Rageau, Mauget, 1999). The reduced proportions of spurs are another indication of sporadic renewal of growth, which leads to cherry low production (Oukabli, Mahhou, 1997; Oukabli, Laghezali, 2000). All these symptoms are an indication of inadequate chilling which limits the performance of sweet cherry under climatic conditions at low elevations in Morocco. Nevertheless, characterization of dormancy in this species was carried out only by few researchers (Arias, Crabbé, 1975), but never under warm climates. The test of « single node cuttings » provides some interesting indications and has constituted an experimental approach for several studies (Champagnat, 1989), especially in locations with warm winters (Balandier et al., 1992; Zguigal, 1995; Zguigal et al., 2006).
3On the other hand, anatomical approach was used to examine the internal structures and to complete biological and morphological observations of the buds for numerous species of Prunus. This technique has revealed that during dormancy, vascular tissues are not completely differentiated and that the connection between floral primordia and the bud axis is ensured only by procambial cells (Ashworth, Rowse, 1982; Ashworth, 1984). This vascular connection appears to be functional one month prior to anthesis in the European plum (Hanson, Breen, 1985). For normal bud break in apricot, it must be established near anthesis (Bartolini, Giorgelli, 1994). The appearance of the tetrades stage during microsporogenis constitute also an indication of a regular release from dormancy (Guerriero, Bartolini, 1991). Therefore, the environmental conditions seem to have a considerable impact on floral differentiation and play a regulatory role of this event.
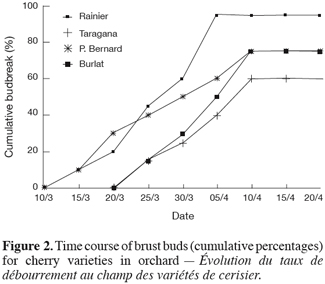
4The objective of this study was to evaluate the behaviour of sweet cherry based on its phenological stages progress and on the evolution of its dormancy and the effects of inadequate chilling on flower morphogenesis during warm winter.
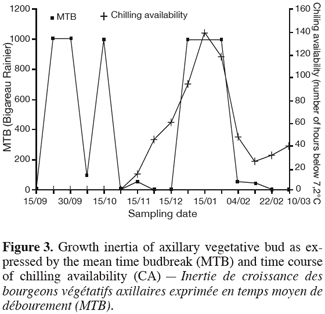
2. Material and methods
2.1. Plant material
5A cherry collection was established in 1983 at Aïn Taoujdate (altitude 500 m) in the experimental station of INRA. Trees were grafted on Saint Lucie 64 rootstock, planted at 5 x 6 m spacing (333 trees per hectare) and trained on an open vase system. They were surface irrigated from May to September with 2000 m3 per year and fertilized with 50-30-80 units per hectare of N-P-K respectively.
2.2. Climatic characterization of the site
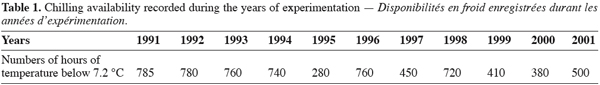
6To evaluate the behaviour of sweet cherry under the environmental conditions, chilling hours (CH) were estimated for the period of experimentation (1991–2001) using a linear model (CH: (7.2-m)/(M-m) x 24 (Weinberger, 1950), where M is the maximum and m the minimum temperature at the experimental site).
2.3. Phenological characterization
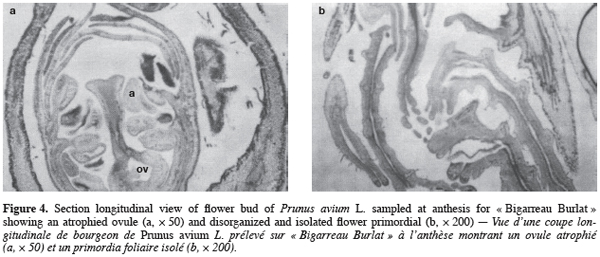
7Progress of phenological stages was undertaken on the tree in collection during five years (1991–1995) and concerned leaf fall, bud break and flowering period. Leaf fall was noted when tree have lost 95% of their leaves. This state was appreciated visually on three trees for each variety. The evolution of buds was recorded on a sample of five, one-year-old, shoots per tree (15 shoots/variety) chosen around the canopy of the trees grown outdoors. These field observations were used to calculate bud break rate by dividing the number of buds broken by the total number of buds. Bud break was considered happening when 50% of buds are at the stage A of Flekinger. Flowering periods were also notified, at the H stage of Flekinger, when the first flowers were opened and full bloom when 50% of flowers are open. The end of flowering corresponds to 90% of flowers without petals. Bud break rate of varieties in the field were compared statistically and means were separated by the Duncan’s test.
2.4. Single node cuttings test
8Study of dormancy evolution was carried out using the single node cuttings (Rageau et al., 1996). Twigs of 20-30 cm length were sampled weekly from trees of four varieties : Burlat, Precoce Bernard, Rainier and Tragana d’Edesa, starting from September (1995) until bud break occurred in the field (April 1996). The shoots were cut into segments of 3 cm of length bearing a single node with a sample of 20 cuttings per variety and per date. The tips of the cuttings were covered with paraffin to limit water loss. The samples were then forced under constant conditions of: temperature of 20°C, photoperiod of 16 hours and 90% of relative humidity. The date of bud break (visible tips of young leaves) was recorded during every 2 days during one month and the mean time of bud break (MTB) in days was calculated according the formula:
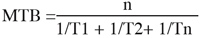
9where n: total buds
10Tn: time in days elapsed prior to bud break
11For sampling dates where the bud break was nil, the corresponding MTB was attributed an arbitrary value of 1000 in order to show periods of important inertia (Rageau, personal communication). The variations in MTB among samples were interpreted in terms of evolution of growth capacity of the buds (Rageau, 1978). The time which elapses until bud break provides a good estimation of the intensity for intrinsic growth inhibition of the bud (endodormancy) (Champagnat, 1989). The increase of MTB translates therefore an increase in the inertia bud growth.
2.5. Histological bud observation
12Anatomical study was carried out on Burlat in order to determine the effects of the lack of chilling on bud differentiation. Thus, twenty flower buds were randomly collected on spurs on the 25th of March 2001 corresponding to anthesis stage. At this date the buds had accumulated about 500 hours of temperatures inferior to 7.2°C. The buds were brought to the lab and fixed in formaldehyde acetic acid (FAA) (90% ethanol at 50°, 5% acetic acid, 5% formaldehyde, v/v/v). Scales were carefully removed, buds washed in tap water and then dehydrated in an alcohol series, embedded in paraffin and sectioned at thickness of 10 μ. Sections were stained with safranin and fast green according to the technique described for apricot (Bartolini, Giorgelli, 1994).
3. Results and discussion
3.1. Climatic characterization
13The chilling hours recorded yearly vary between 280 and 785 (Table 1) with an average of 595 hours below 7.2°C. The monthly break-up of these chilling amount show that the cold period is short and the continuous chilling was recorded principally between December and February (Table 2). This lack of chilling affects the behaviour of cherry and disrupts the progress of phenological stages and consequently cherry cropping potential.

3.2. Phenological stages
14Varieties tested display different behaviour as revealed by the progress of phenological stages. Leaves on the spurs fall the first ones and fallen leaf was observed during the first week of January with difference among varieties. « Precoce Bernard » and « Rainer » were the first ones that loose leaves (Table 3). The fallen leaves happened when trees were exposed at about 200 hours of chill units. The differences observed among varieties were linked to their sensitivity to the environmental factors notably to short photo-period, low temperatures and thermic amplitudes. Varieties « B. Burlat » and « Tragana d’Edessa » presented an important delay among years and translate their sensitivity to change in climatic factors.
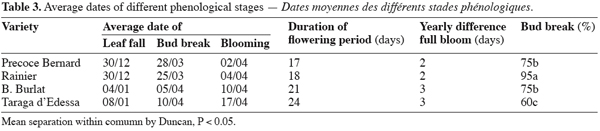
15Bud break was not linked to date of fallen leaves. This stage was observed during the last week of March (Table 3) for « Rainier » and « Précoce Bernard » and the first week of April for the others two varieties. The progress of this stage was late and heterogeneous for « B. Burat » and « Tragana d’Edessa » related to their high requirements of low temperatures. Vegetative growth started from the terminal buds, and apical dominance was marked.
16Flowering stage was observed in the first days of April for « Précoce Bernard » and « Rainier » and at the second decade of April for the other varieties. The first flowers opened are those carried by the spurs (Bouquet de Mai). They constitute the first flowering flux followed by the flowers carried on the base of one year shoot but which were not numerous. The flower index was low for « B. Burlat » and « Tragana d’Edessa » and flowers do not often set fruit, indicating an inadequate chilling duration for flower development and lack of climatic adaptation.
3.3. Evolution of bud dormancy
17The curves of growth inertia (TMB) of sweet cherry vegetative bud, show different shapes (Figure 1a and b). This variability indicates the existence of differential behaviour of varieties in response to environmental conditions. Buds are submitted to two periods of important inertia: a period of probably correlative inhibition then a period of winter dormancy. The first one is situated between the end of September and the middle of October. The second phase lies between the end of October to the end of February or early March, depending on the variety. In fact, the growth capacity becomes important for « Rainier » and « Precoce de Bernard » starting by end of February and the first decade of March, but remains low for « B. Burlat » and very low for « Tragana d’Edessa ».
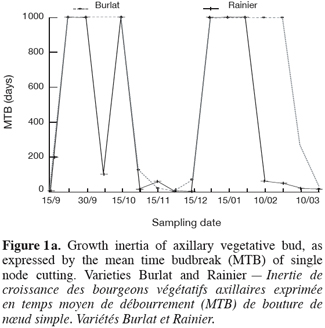
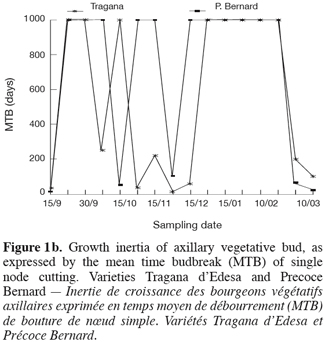
18The correlative inhibition decreases as the leaves drop and as the autumnal low temperatures arrive. The buds go into a period of dormancy with variable intensity. The removal of such dormancy exhibits different patterns and occurs at different rhythms depending on the variety. Thus, the acquisition of the capacity of bud break is relatively rapid for « Rainier » and « Précoce Bernard », slow for « Burlat » and very slow for « Tradana d’Edessa ». For the last two varieties, bud break in orchard was incomplete (low rate) and spread over time (Figure 2) which constitutes an indication of the lack in chilling.
19Dormancy (second period on inertia) has begun after the buds had accumulated 200 hours of low temperatures. During this phase, the amount of chilling received was 400 hours which seem to be insufficient to overcome dormancy especially when the autumnal chilling plays a major role in the onset of dormancy (Figure 3). Similar results have been reported on apple in the same region (Zguigal, 1995; Zguigal et al., 2006). The classical quantification of chilling (as a linear process) leads therefore to an overestimation of available chilling that is effective in the overcome of dormancy. In order to correct this discrepancy it is recommended to take into consideration:
20– the chilling hours accumulated mainly during the second inertia period;
21– high and moderate temperatures inter-dispersed during the dormancy period (duration and frequency);
22– reconsider temperatures thresholds used in the quantification of chilling instead of 7.2°C which is not necessarily appropriate for locations with warm winter.
23The inhibition of bud growth revealed by the single node cuttings could be due to the dormancy of the bud sensu stricto (endo-dormancy) and/or to correlative inhibition (Rageau et al., 1996). It is worthy to notice that dormancy in Meknes area (mild winter) shows an inertia periods which last longer than in Belgium (temperate climate) (Crabbé, 1994).
24Differences in physiological stages of buds exist between the first and the second period. During the first period, correlative inhibitions from spring and summer shoot (leaves) continue to exert their effect on the buds. In the second period, leaves abscission and the arrival of low temperatures play an important role in the onset of dormancy and the decrease of inertia. However, a delay in the decrease of this inertia is translated by an incomplete and low rate of bud break.
3.4. Organogenis flower and xylem vessel state
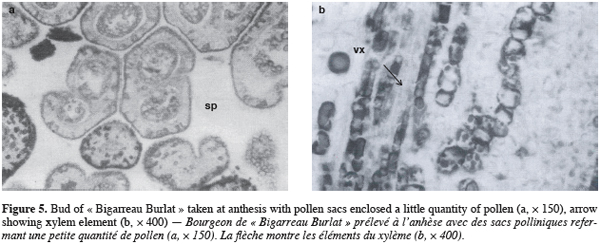
25Anatomical observations of flower buds at anthesis revealed that reproductive organs showed different abnormalities. Flowers present malformations with some having no pistil. When the latter is present, the ovary is always of a small size (Figure 4a). The female gametophyte presents an atrophied ovule, isolated in the ovary cavity. Nucellus tissue is separated from the teguments which constitutes an indication of ovular degeneration. Abnormalities in differentiation were also observed (Figure 4b) with a disorganized and interlaced flower primordia.
26Anthers contain pollen sacs, which remained indehiscent. The layers were not separated and sacs contain cells in tetrad stage (Figure 5a). At bud break the microsporogenis was low, and pollen mother cells were immature, many of which show signs of abortion like wall disorganization. If meiosis has occurred, even under insufficient chilling, the reduction in amount of pollen maturation would limit the possibilities of pollination of self-incompatible Burlat. Flower organs (male and female) were thus characterized by various abnormalities, which indicate a perturbation of the flower differentiation phase. On the other hand, anatomical observation showed xylem vessel elements at the bases of buds and flower primordia, mainly anther and pistil (Figure 5b). This suggests that vascular connection is usually established during dormancy and becomes fully functional during the flowering period, as it was reported for other species of Prunus (Ashworth, Rowse, 1982; Hanson, Breen, 1985). Even though water conductivity is apparently ensured in the bud axis and flower organs for a normal break, the subsequent evolution of the buds remains sporadic with a number of them dropping during or after flowering.
27The introduction of sweet cherry cultivars like « Burlat » in areas with mild winters leads to low yields. The low productivity is due largely to the lack of chilling leading to low rate and sporadic bud break in addition to anatomical abnormalities in flower buds. Similar results were reported on apricot (Rageau et al., 1996) and apple (Oukabli et al., 2003). In this process, the impact of autumnal climatic conditions should not be ignored. In fact, these conditions play an important role in flower differentiation and floral organogenis just as do moderate and high temperatures (Oukabli, Mahhou, 1997; Oukabli, Laghezali, 2000). Low bud break rate and ovule and pollen abortion led to low production of the variety « Bigarreau Burlat » and for others also. Chilling duration needed to release dormancy is high and varies among cultivars like it was reported on some compatibles ones (Mahmood et al., 2000b). Hence, successful production of cherry under warm climatic conditions (with the absence of adequate winter chilling) is not possible.
Bibliographie
Arias O., Crabbé J. (1975). Les gradients morphogénétiques du rameau d'un an des végétaux ligneux, en repos apparent. Physiol. Végét. 13 (1), p. 69–81.
Ashworth EN. (1984). Xylem development in Prunus flower buds and the relationship to deep supercooling. Plant Physiol. 74, p. 862–865.
Ashworth N., Rowse DJ. (1982).Vascular development in dormant Prunus flower buds and its relationship to supercooling. Hort. Sci. 17 (5), p. 790–791.
Balandier P., Capitan F., Quignot S, Rageau R, Parisot E. (1992). étude de la croissance et du développement des bourgeons du pêcher cultivé à l’île de la Réunion : application à l'élaboration d'un itinéraire cultural. Fruits 47 (6), p. 679–689.
Bartolini S., Giorgelli F. (1994). Observations on development of vascular connections in two apricot cultivars. Adv. Hortic. Sci. 8, p. 97–100.
Champagnat P. (1989). Rest and activity in vegetative buds of trees. In Dreyer and al. (eds). Forest tree physiology. International Symposium, INRA, Université de Nancy, Proceedings, Nancy (France). Ann. Sci. For. 46 (suppl.), p. 9–26.
Crabbé J. (1987). Aspects particuliers de la morphogenèse caulinaire des végétaux ligneux et introduction à leur étude quantitative. Bruxelles, Belgique : IRSI, 116 p.
Crabbé J. (1994). Dormancy. In Arntzen C. (ed.). Encyclopedia of Agricultural Science. Vol.1. New York: Academic Press, p. 597–611.
Guerriero R., Bartolini S. (1991). Main factors influencing cropping behaviour of some apricot cultivars in coastal areas. Acta Hortic. 293, p. 229–243.
Hanson EJ., Breen PJ. (1985). Xylem differentiation and boron accumulation in « Italian » prune flower buds. J. Am. Soc. Hortic. Sci. 110 (4), p. 566–570.
Mahmood K., Carew JG., Hadley P., Battey NH. (2000a). The effect of chilling and post-chilling temperatures on growth and flowering of sweet cherry (Prunus avium L.). J. Hortic. Sci. Biotechnol. 75 (5), p. 598–601.
Mahmood K., Carew JG., Hadley P., Battey NH. (2000b). Chill unit models for swet cherry cvs Stella, sunburst and Summit. J. Hortic. Sci. Biotechnol. 75 (5), p. 602–606.
Oukabli A., Mahhou A. (1997). Abondance des bouquets de mai et induction florale chez le cerisier, Prunus avium L. en climat marocain à hiver doux. Fruits 52, p. 117–181.
Oukabli A., Laghezali M. (2000). Evaluation phénologique et pomologique d’une collection variétale de cerisiers en conditions de moyenne altitude. Fruits 55 (1), p. 83–92.
Oukabli A., Bartolini S., Viti R. (2003). Anatomical and morphological study of apple (Malus x domestica Borth.) flower buds growing under inadequate winter chilling. J. Hortic. Sci. Biotechnol. 78 (3), p. 580–585.
Oukabli A., Mamouni A., Laghezali M. (2002). Contribution à l’étude des causes histologiques du manque de fructification chez le cerisier doux (Prunus avium L.) conduit en zone de moyenne altitude. Al-Awamia 103, p. 39–49.
Rageau R. (1978). Croissance et débourrement des bourgeons végétatifs de pêcher (Prunus persica L. Batsch). C. R. Acad. Sciences. Paris, Série D. Sci. Nat. 226 (18), p. 1845–1848.
Rageau R., Julien JL., Ollat N. (1996). Approches du contrôle de la croissance des bourgeons dans le contexte de l’arbre entier (107–120). In Cruiziat P., Lagouarde JP. (eds.). Actes de l’école chercheurs Bioclimatologie, INRA, 3-7 Avril 1995, Le Croisic, France. Tome 1: De la plante au couvert végétal. Paris : INRA Editions, p. 107–120.
Rageau R., Mauget JC. (1999). Dormancy dynamics and phenology in peach, apple and walnut trees under prolonged mild winter temperatures in a heated greenhouse. Symposium, ISHS. Acta Hortic. 199, p. 71.
Zguigal Y. (1995). Evolution et caractéristiques de la dormance des bourgeons du Pommier (Malus x domestica Borkh.; cv Golden Delicious) dans un climat à hiver doux (région de Meknes, Maroc). Thesis. Rabat, Marocco: Institut agronomique et vétérinaire Hassan II.
Zguigal Y., Chahbar A., Wallali-Loudiyi DE., Crabbé J. (2006). Caractéristiques de la dormance des bourgeons du pommier dans les régions à hiver doux. Biotechnol. Agron. Soc. Environ. 10 (2), p. 131–137.
Weinberger JH. (1950). Chilling requirements of peach varieties. Proc. Amer. Soc. Sci. 56, p. 122–128.
Pour citer cet article
A propos de : Ahmed Oukabli
Institut national de la Recherche agronomique. Amélioration des plantes et Conservation des ressources phytogénétiques. CRRA, BP 578, Meknes (Maroc). E-mail : oukabli2001@yahoo.fr
A propos de : Ahmed Mahhou
Institut agronomique et vétérinaire Hassan II. Département d’Horticulture. BP 6202, Rabat (Maroc).





