- Accueil
- Volume 15 (2011)
- numéro spécial 1
- Modeling cadmium in the feed chain and cattle organs
Visualisation(s): 3505 (10 ULiège)
Téléchargement(s): 93 (1 ULiège)
Modeling cadmium in the feed chain and cattle organs

Abstract
The objectives of this study were to estimate cadmium contamination levels in different scenarios related to soil characteristics and assumptions regarding cadmium accumulation in the animal tissues, using quantitative supply chain modeling. The model takes into account soil cadmium levels, soil pH, soil-to-plant transfer, animal consumption patterns, and transfer into animal organs (liver and kidneys). The model was applied to cattle up to the age of six years which were fed roughage (maize and grass) and compound feed. Cadmium content in roughage and cadmium intake by cattle were calculated for six different (soil) scenarios varying in soil cadmium levels and soil pH. For each of the six scenarios, the carry-over of cadmium from intake into the cattle organs was estimated applying two model assumptions, i.e., linear accumulation and a steady state situation. The results showed that only in the most extreme soil scenario (cadmium level 2.5 mg.kg-1, pH 4.5), cadmium exceeded the EC maximum tolerated level in roughage. Assuming linear accumulation, cadmium levels in organs of cattle up to six years of age, ranged from 0.37-4.03 mg.kg-1 of fresh weight for kidneys and from 0.07 to 0.77 mg.kg-1 of fresh weight for livers. The maximum tolerated levels in one or both organs were exceeded in several scenarios. When considering organ excretion of cadmium, internal cadmium levels in organs were approximately one order of magnitude lower as compared to the results of the linear accumulation model. In this case only in the most extreme soil scenario, the maximum tolerated level in the kidney was exceeded. It was concluded that the difference between the two assumptions (linear model versus a steady state situation to estimate cadmium carry-over in cattle) is negligible in the animal’s first five years of life, but will become relevant at higher ages. For the current case, the linear approach is a good descriptor for worst case situations. Furthermore, this study showed that quantitative supply chain modeling is an effective tool in assessing whether or not a specific combination of soil properties would lead to unacceptable contaminant levels in feedstuffs and animal products in the view of animal and human health.
Table des matières
1. Introduction
1The supply of safe feed products to animals is crucial not only to safeguard animal health and welfare but also to reduce human exposure to potentially toxic compounds (PTC) like heavy metals and organic contaminants. Carry-over of PTC from feed into consumable animal products, like liver, kidney and muscles (meat), can contribute substantially to human intake of these compounds, particularly in case of heavy metals like cadmium. Therefore, legislative maximum limits have been set within the EU for a number of PTC, including heavy metals, in animal feedstuffs (2002/32/EC) and animal derived food products (2001/466/EC). To comply with these limits, control in every stage of the feed supply chain is needed, starting at the initial source of contamination and covering all other relevant stages of the feed chain. The number of relevant processes and stages depends on the particular contaminant of concern. For heavy metals, “control” already starts at the soil and/or water resources. Obviously, intensive monitoring of the safety of all parts of the feed and food chain, from the soil up to the feedstuffs and animal derived food products, is very cost- and labour-intensive. Knowledge of transfer processes of specific PTC through the feed and food chain offers a way to optimize monitoring activities and/or reduce their costs. Supply chain models, thereby, can help the development of intervention measures to control the contamination of PTC in the final feed and food products and can be used to assess the effectiveness of these control measures (Römkens et al., 2008; Van der Fels-Klerx et al., 2008; van Raamsdonk et al., 2009). Obviously, such a modeling approach should include all stages of the supply chain of interest, including different contamination pathways and carry-over coefficients.
2Heavy metals, such as cadmium (Cd), zinc (Zn), copper (Cu) and lead (Pb), are a group of PTC that are of concern when dealing with the quality of animal feedstuffs, particularly roughage. In the Kempen area of the Netherlands as well as in other European countries diffuse pollution of Cd has resulted in elevated levels in the soil. The presence of Cd in soils used for the production of feedstuffs results in high levels in the feed product due to uptake by the plant and consecutive accumulation in edible plants parts and in animals (Rietra et al., 2007). The degree to which Cd is available for plant uptake and further transfer into the feed chain strongly depends on the degree of pollution and soil characteristics. Hence, regional differences in soil Cd levels and soil characteristics will result in differences in contamination levels of plants used for feed production. In the animal’s body, Cd may accumulate in the organs and/or be excreted by products like milk, meat and eggs and by the animal’s metabolism. The extent of deposition and elimination in the animal depends on the half-life time of the compound and the period considered.
3This study aimed to use supply chain modeling to estimate Cd levels in animal feed and animal organs in different scenarios related to soil characteristics and assumptions regarding Cd accumulation in the animal. As a case, the model was applied to cattle aged up to six years, which were fed roughage and compound feed.
2. Materials and methods
2.1. Model overview and scenarios
4A supply chain model that links Cd in the soil to Cd in animal organs has been developed with the aim to estimate Cd levels in these animal derived food products. The model takes into account soil Cd levels, soil pH, soil-to-plant transfer, animal consumption patterns, and transfer into liver and kidneys. A schematic overview of the model is presented in figure 1.
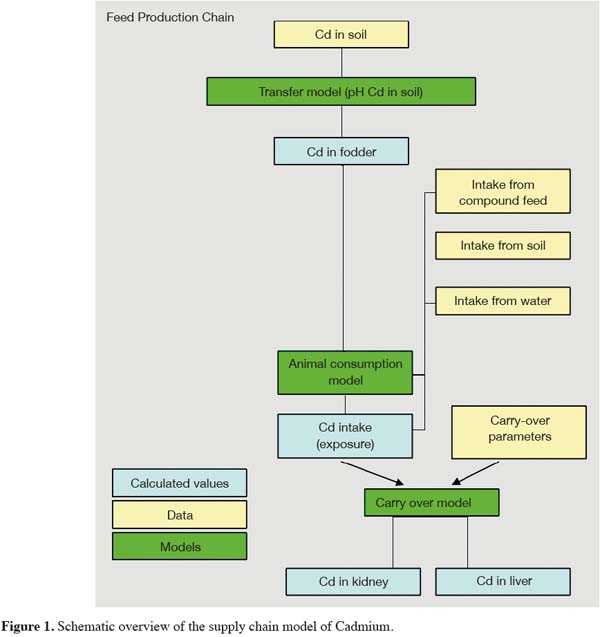
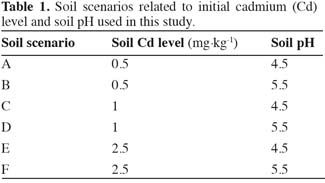
5The model was applied to cattle from 0 to 6 years of age, which were fed roughage, including maize and grass, and compound feed. Cadmium content in roughage and Cd intake by cattle were calculated for six different soils, as presented in table 1. These soil scenarios reflect the range of Cd in the soil and soil pH for acid sandy soils in the Kempen area in The Netherlands. The elevated Cd levels in soil are due to diffuse pollution from Cd and Zn smelters both in The Netherlands and in Belgium.
6Two different model approaches were applied to calculate the carry-over of Cd from intake into the cattle organs. The first one is a linear (irreversible) accumulation model which calculates the final Cd level in the organs based on a linear bioconcentration factor considering the total accumulated Cd intake. The second one is a non-linear steady-state model that also considers excretion of Cd and the development of a steady state situation. By definition, daily Cd intake equals daily elimination by metabolism and/or by excretion at steady-state. Differences between the two carry-over models were compared in terms of final Cd levels in the cattle organs (liver, kidney) maintaining all other model inputs equal. The two carry-over models were applied to each soil scenario, resulting in 12 different scenarios.
2.2. Model description
7The supply chain model consists of two modules: the soil-plant module and the animal module. The soil-plant module calculates Cd concentrations in plants based on the soil Cd level, soil pH, organic matter and clay content. The animal module first calculates the total daily and annual Cd intake by animals considering the intake of roughage (grass and maize), compound feed, water and soil. Differences in the consumption patterns of younger and older cows are considered. Subsequently, the levels of Cd in liver and kidney are calculated based on either the linear bioconcentration model assuming accumulation only or the non-linear accumulation-excretion model. Below a summary of both modules is given; more details can be found in Franz et al. (2008) and Römkens et al. (2008).
8Soil-plant module. First of all, Cd levels in grass and maize were calculated using an extended Freundlich equation that considers differences in Cd in soil, organic matter, clay and soil pH according to:
9log[Cdplant] = Constant + a·log[organic matter] + b·log[clay] + c·log[Cdsoil] + d·[pH] (1)
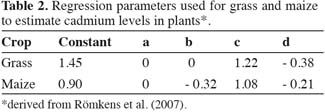
10with both Cd levels in the plant (Cdplant) and Cd levels in the soil (Cdsoil) expressed in mg.kg-1 of dry matter. Freundlich equations have been used previously to successfully predict metal levels in crops like wheat (Adams et al., 2004) and rice (Römkens et al., 2009). Both organic matter and clay are expressed as percentage, and were fixed at 3% based on measurements in the Kempen area. Cdsoil and pH were grouped into the six scenarios presented in table 1, and are representative for non-polluted, slightly polluted and moderately polluted soils typically for this region. The soils are predominantly sandy with a low clay and organic matter content. The parameters of the regression coefficients a, b, c, and d were estimated based on regression analyses of field data (Römkens et al., 2007). The resulting coefficients used in equation 1 are presented in table 2.
11Animal module
12Animal intake. The daily Cd intake of cattle (DI in mg Cd per day) was calculated as the sum of the intake of soil, roughage (including grass and maize in a fraction of 70 to 30%), and compound feed, each of the three multiplied by their respective Cd contamination levels, see Equation (2). The intake of water was proven to be negligible based on measurements of Cd in drinking water (Römkens et al., 2007).
13DI = ∑ (Cdsoil x Cosoil) + (Cdcompound x Cocompound) + (Cdroughage x Coroughage) (2)
14Cd levels in the soil (Cdsoil), in compound feed (Cdcompound) and in roughage (Cdroughage) are expressed in mg per kg dry matter. Cosoil, Cocompound and Coroughage represent the animal daily consumption of respectively soil, compound feed and roughage, expressed in kg dry matter per day. Soil ingestion (Cosoil) was assumed to be due to soil attached to roughage and was fixed at the level of 3% of total grass and maize consumption. Soil Cd contamination levels (Cdsoil) were based on the six scenarios (Table 1).
15In The Netherlands, raw material for the production of compound feed is mainly imported, rather than produced locally. Therefore, levels of Cd in compound feed (Cdcompound) were based on monitoring results as stored into a database with the results of national monitoring programs on chemical contamination levels in feed and food products and their commodities (Van Klaveren, 1999). Based on this database, the median Cd level in compound feedstuffs was 0.05 mg per kg dry matter. Cadmium levels in grass and maize (Cdroughage) were estimated using the soil-plant module.
16The average daily intake of cattle was calculated for three age groups, being: 0-1 year, 1-2 years and > 2 years. Intake of grass, maize and compound feed in these three age groups was based on Römkens et al. (2007). For total roughage intake (Coroughage), it was estimated to be 4, 8, and 14 kg dry matter per day. This resulted in an average daily intake for the three age groups of, respectively, 0.12, 0.24 and 0.42 kg of dry matter per day for soil; 2.8, 5.6 and 9.8 kg of dry matter per day for grass; and 1.2, 2.4 and 4.2 kg of dry matter per day for maize. Consumption of compound feed (Cocompound) was estimated to be 0.24, 0 and 2.32 kg of dry matter per day for the three age groups, respectively.
17Carry-over. Carry-over of Cd to cattle animal organs (kidney and liver) was modeled according to two different approaches: one based on linear accumulation in target organs and one assuming the development of a steady state situation using an exponential accumulation-excretion model (Römkens et al., 2008). In case of linear accumulation, Cd irreversibly accumulates in the cattle liver and kidneys; there is no excretion from these target organs. This has been supported by several studies on Cd accumulation in organs of sheep (Loganathan et al., 1999) and cattle of 2.5 to 8 years of age (Olsson et al., 2001). The accumulation-excretion hypothesis on the other hand is also supported by several studies, e.g. by those for Cd in kidneys of cattle (Smith et al., 1991; Underwood et al., 1999). It has been suggested also that accumulation and excretion depend on the level of exposure or the age of the animals. For example, lower excretion rates were observed in sheep at an age between 3 and 28 months at higher exposure levels (Lee et al., 1996). Data of Spierenburg et al. (1988) for cattle can be described with a linear model until an age of up to five years old (Römkens et al., 2008), but at higher ages the accumulation levels tend to decrease, which favors the application of an exponential model. Excretion of Cd by urine was also found in cattle, e.g. by Smith et al. (1991).
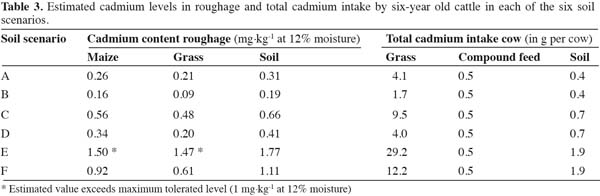
18The irreversible transfer of Cd into kidneys and liver in the linear accumulation model is calculated using a biotransfer rate (BTR). The BTR is defined as the increase of the Cd concentration in the organ tissue per day divided by the additional Cd intake per day, and expressed per 1 kg of tissue. Rates for the BTR used are 9.0 x 10-5 for kidneys and 1.7 x 10-5 for liver (Franz et al., 2008). The linear BTR model applied is represented in Equation (3):
19Ct = BTR × DI × t (3)
20with Ct representing the Cd concentration in the organ (in mg.kg-1) after a period of t (days).
21In case of the development of a steady state situation, Cd not only accumulates in the target organs but is excreted as well. In this case, the relationship between Cd intake and Cd levels in the organs is not linear but described by an exponential model. For all routes (accumulation and elimination) the concentration at day t can be described as follows:
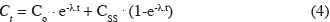
22For which applies:
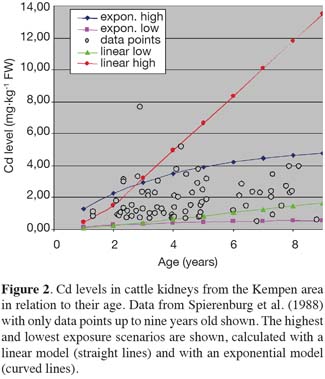
23CSS = BTF . D (5)
24and
25D = DI × Fabs
26With Ct: concentration of contaminant (mg.kg-1) at day t, C0: concentration of contaminant (mg.kg-1) at day 0, the starting level of the simulated period of time, CSS: steady state concentration (mg.kg-1), λ: elimination time constant (1 per day), to be calculated from the half life time T1/2, with λ = ln (2) / T1/2
27BTF: biotransformation factor (day.kg-1), D: daily total uptake (mg.day-1), Fabs: absorption in the alimentary canal (%).
28The BTF in (5) is defined as the constant to calculate the steady state concentration (mg.kg-1) from the uptake (mg.day-1) and has, therefore, the unit day.kg-1. The biotransformation factor BTF for accumulation in an organ is calculated as follows:

29with COR: carry-over rate to the target organ (no dimension), W: weight of the organ (kg), λ: elimination time constant (1 per day).
30In this study, an absorption factor (Fabs) of 20% (van Raamsdonk et al., 2007), a carry-over rate (COR) of 0.079% and a half-life time (T1/2) of 900 days for kidneys was applied (van Raamsdonk et al., 2007). Comparable carry-over rates are reported by Neathery et al. (1975) and Kreuzer (1986). Adjusted to an absorption factor of 100%, the COR equals 16 x 10-5 (0.016%), which is comparable to the BTR used.
3. Results
31Estimated Cd levels in roughage and the estimated total Cd intake of cattle are presented in table 3 for each of the six soil scenarios. The estimated Cd levels in roughage ranges between 0.16 and 1.50 mg.kg-1 at 12% moisture for maize and between 0.09 and 1.47 mg.kg-1 at 12% moisture for grass, depending on the soil scenario. Only in the most extreme scenario (scenario E: Cd soil level of 2.5 mg.kg-1 and pH of 4.5), Cd in roughage exceeded the EC maximum tolerated level (1 mg.kg-1 at 12% moisture, see 2002/32/EC). The total Cd uptake by the cow is dominated by grass consumption in each scenario.
32The estimated Cd levels in the cattle organs are presented in table 4 for each of the six soil scenarios and the two carry-over scenarios. Assuming linear accumulation during a life span of six years, Cd levels in cattle organs range from 0.37-4.03 mg.kg-1 of fresh weight (FW) for kidneys and from 0.07 to 0.76 mg.kg-1 of FW for livers. The maximum tolerated levels for both organs were exceeded in the most extreme soil scenario (scenario E). The maximum tolerated level in kidneys was also exceeded in two other scenarios (scenarios C and F).
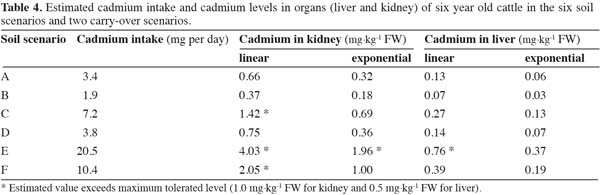
33Assuming excretion from the organs, Cd levels in the cattle organs after six years are approximately a factor two lower as compared to applying a linear model. Only in the most extreme scenario (scenario E) the maximum level in the kidney was exceeded.
4. Discussion and conclusion
34The difference between the two approaches, linear versus exponential modeling in estimating carry-over of Cd by cattle is small during the animal’s first five years, but will become relevant at higher ages. Since we consider a cattle productive life span of six years which is about two times the contaminant biological half-life time (900 days), the linear approach is a good descriptor for worst case situations in this study. However, for those situations where estimated levels of cadmium equal or exceed legal limits, more precise estimations are required.
35It can be calculated that, approximately, 97% of the steady-state-level is reached after five times the half-life time of the contaminant. In the case of Cd this situation will never been reached, because of the assumed (very) long half-life time.
36The half-life time for Cd in kidneys as used in the exponential model results from fitting the model to the data of Spierenburg et al. (1988), see figure 2. Using a BTR of 2 × 10-4 (Römkens et al., 2008) or a COR of 0.12% (absorption factor of 20%, see van Raamsdonk et al., 2007), linear and exponential models describe the data of Spierenburg et al. (1988) equally well for all data points younger than five years. Therefore the most simple, i.e. linear model, can be chosen. The difference between the models increases rapidly at higher ages. Olsson et al. (2001) fitted a linear relationship between cattle age and Cd levels in the kidney, with age running up to 12 years. They found a yearly increase of about 26 mg.kg-1 Cd in the kidney. However, the percentage of explained variance of the linear model was low, which may have been due to data related to the older ages. An exponential approach will give a more realistic description at cattle above five years of age.
37This study shows that quantitative supply chain modeling is an effective tool to estimate contaminant levels in feedstuffs and animal products. It enables the user to assess whether or not a specific combination of soil properties, soil acidity and land use leads to unacceptable levels of animal exposure in view of product quality and human health. The model can be used for a relatively fast evaluation of cadmium exposure for specific regions, without the necessity of setting up extensive soil and crop monitoring schemes. Based on initial model assessment, monitoring can focus on those areas where product quality is expected to exceed legal limits for Cd in both animal fodder and products for human consumption. Due to the limited number of model parameters the model is also easy to adapt to other regions and/or other contaminants. This facilitates the harmonization of risk assessment procedures considerable and will help to overcome apparent differences in legal limits in soil which do exist at present.
38Acknowledgements
39This study was financed by INTERREG III and the Dutch Ministry of Agriculture, Nature and Food Quality. This paper is based on a presentation given at the 3rd International Feed Safety Conference – Methods and Challenges (joint to Cost Action FA0802), held 6-7 October 2009 in Wageningen, The Netherlands, and was supported by Feed for Health, COST Action FA 0802 (www.feedforhealth.org).
Bibliographie
Adams M.L. et al., 2004. Predicting cadmium concentrations in wheat and barley grain using soil properties. J. Environ. Qual., 33(2), 532-541.
European Commission, 2002. Directive 2002/32/EC of the European Parliament and of the Council of 7 May 2002 on undesirable substances in animal feed. Off. J. Eur. Community, L140, 10-21.
European Commission, 2006. Commission regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union, L364, 5-24.
Franz E., Römkens P., van Raamsdonk L. & Van der Fels-Klerx H.J., 2008. A chain modeling approach to estimate the impact of soil cadmium pollution on human dietary exposure. J. Food Prot., 71(12), 2504-2513.
Kreuzer W., 1986. Zum Übergang von Cadmium bei Mast- und Nutztieren. In: Hecht H., ed. Zum Carry-over von Cadmium. Münster-Hiltrup, Germany: Landwirtschaftsverlag GmbH.
Lee J., Roince J.R., Mackay A.D. & Grace N.D., 1996. Accumulation of cadmium with time in Romney sheep grazing ryegrass-white clover pasture: effect of cadmium from pasture and soil intake. Aust. J. Agric. Res., 47, 877-894.
Loganathan P. et al., 1999. A model to predict kidney and liver cadmium concentrations in grazing animals. New Zealand J. Agric. Res., 42, 423-432.
Neathery M.W. & Miller W.J., 1975. Metabolism in animals of cadmium, mercury, and lead as related to their toxicology and ecology. In: Proceedings of the Georgia Nutrition Conference. Athens: University of Georgia, 65-80.
Olsson I.-M., Jonsson S. & Oskarsson A., 2001. Cadmium and zinc in kidney, liver, muscle and mammary tissue from dairy cows in conventional and organic farming. J. Environ. Monit., 3, 531-538.
Rietra R.P.J.J. & Römkens P.F.A.M., 2007. Cadmium and zinc in soil and grass from nature areas in the Kempen area. Alterra report 1497 (in Dutch). Wageningen, the Netherlands: Alterra-Wageningen UR.
Römkens P.F.A.M. et al., 2007. Exposure to cadmium and intake by cattle in the Kempen area: modelling results. Alterra report 1438. Wageningen, The Netherlands: Alterra-Wageningen UR.
Römkens P. et al., 2008. Geoquality - Part I. Chain models as a tool to quantify the relation between soil, crop quality and human exposure. In: Hamer & Petersen, eds. From stable to table - Food safety and quality in international food chains. Düsseldorf, Germany: MUNLV; Bonn: GIQS, 198-218.
Römkens P.F.A.M. et al., 2009. Prediction of Cadmium uptake by brown rice and derivation of soil-plant transfer models to improve soil protection guidelines. Environ. Pollut., 157, 2435-2444.
Smith R.M. et al., 1991. Effects of long-term dietary cadmium chloride on tissue, milk, and urine mineral concentrations of lactating dairy cows. J. Anim. Sci., 69, 4088-4096.
Spierenburg Th. J. et al., 1988. Cadmium, zinc, lead, and copper in livers and kidneys of cattle in the neighbourhood of zinc refineries. Environ. Monit. Assess., 11, 107-114.
Underwood E.J. & Suttle N.F., 1999. The mineral nutrition of livestock. 3rd ed. Wallingford, UK: Cabi Publishing.
van der Fels-Klerx H.J., Tromp S., Rijgersberg H. & Van Asselt E.D., 2008. Application of a transmission model to estimate performance objectives for Salmonella in the broiler supply chain. Int. J. Food Microbiol., 128(1), 22-27.
Van Klaveren J.D., 1999. Quality programme for agricultural products. Results residue monitoring in The Netherlands. Technical report. Wageningen, The Netherlands: RIKILT-Institute of Food Safety.
van Raamsdonk L.W.D. et al., 2007. Schadelijke stoffen in de dierlijke productieketen: overdracht gemodelleerd in ketenperspectief. RIKILT report 2007-2009 (in Dutch). Wageningen, The Netherlands: RIKILT-Institute of Food Safety.
van Raamsdonk L.W.D. et al., 2009. Compliance of feed limits, does not mean compliance of food limits. Biotechnol. Agron. Soc. Environ., 13(S), 51-57.
Pour citer cet article
A propos de : Ine van der Fels-Klerx
RIKILT. Wageningen University and Research Centre. PO Box 230. NL-6700 AE Wageningen (The Netherlands). E-mail: ine.vanderfels@wur.nl
A propos de : Paul Römkens
ALTERRA. Wageningen University and Research Centre. PO Box 47. NL-6700 AA Wageningen (The Netherlands).
A propos de : Eelco Franz
RIKILT. Wageningen University and Research Centre. PO Box 230. NL-6700 AE Wageningen (The Netherlands) – National Institute for Public Health and the Environment. PO Box 1. NL-3720 BA Bilthoven (The Netherlands).
A propos de : Leo van Raamsdonk
RIKILT. Wageningen University and Research Centre. PO Box 230. NL-6700 AE Wageningen (The Netherlands).





