- Accueil
- volume 18 (2015)
- number 1
- Description of Kettneraspis? prescheri sp. nov. (Trilobita, Odontopleuridae) from the “couche rouge” (Pragian, Lower Devonian) in Morocco
Visualisation(s): 7680 (85 ULiège)
Téléchargement(s): 1224 (17 ULiège)
Description of Kettneraspis? prescheri sp. nov. (Trilobita, Odontopleuridae) from the “couche rouge” (Pragian, Lower Devonian) in Morocco

Résumé
Description de Kettneraspis? prescheri sp. nov. (Trilobita, Odontopleuridae) dans la «couche rouge» (Praguien, Dévonien inférieur) du Maroc. L’espèce Kettneraspis? prescheri sp. nov. est décrite dans la Formation Ihandar à Jbel Issoumour, SE du Maroc. Les relations de Kettneraspis Prantl & Přibyl et Leonaspis Richter & Richter, présentant une morphologie similaire, sont discutées. Les diagnoses précédemment publiées ne sont plus considérées comme valides.
Abstract
Kettneraspis? prescheri sp. nov. is described from the Ihandar Formation at Jbel Issoumour, SE Morocco. Relationships of the morphologically similar Kettneraspis Prantl & Přibyl and Leonaspis Richter & Richter are discussed. Previous diagnoses in the literature are no longer considered to be valid.
Table des matières
1. Introduction
1A new odontopleurid trilobite species from the Lower Devonian of Morocco is described. It is named after Harald Prescher, an outstanding friend and researcher who recently passed away much too soon.
2. Geological context
2The “couche rouge” is an informal term used for a reddish bed in the Ihandar Formation (Pragian) which provides a distinctive marker in the landscape as a result of extensive mining (Fig. 1). The holotype was collected at Jbel Issoumour, a cuesta near Alnif, southern Morocco; its exact provenance at the type locality however, is unknown. The site was visited by Harald Prescher and one of us (HH) in 2010. Reference is made to Morzadec (2001) for more details.

Figure 1. Jbel Issoumour, Morocco. Photo taken by HH in 2010.
3Kettneraspis? prescheri sp. nov. is restricted to the Ihandar Formation (“couche rouge”) at Jbel Issoumour and co-occurs here with the probably equally endemic Metacanthina issoumourensis Morzadec, 2001.
3. Systematic palaeontology
4One specimen is housed at the “Institut royal des Sciences naturelles de Belgique” (abbreviated IRSNB) and was treated with ammonium chloride prior to photography; the other is housed at the Forschungsinstitut und Naturmuseum Senckenberg, Frankfurt am Main (SMF), and was treated with magnesium oxide. Morphologic terminology follows Whittington & Kelly (1997). The term “paired axial glabellar tubercles” (Fig. 2) was adopted from Whittington (1956, p. 161, fig. 1). The pygidial major border spines are a pair of large spines proximally connected to a ridge that runs over the pleural lobe and merges with the anterior pygidial axial ring. Adaxially and abaxially to the major border spines are the interior and lateral border spine pairs, respectively.
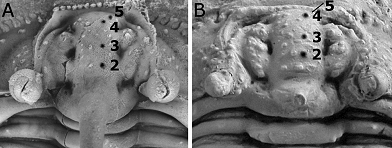
Figure 2. Indications of paired glabellar axial tubercles. A. Kettneraspis? prescheri sp. nov. from the Ihandar Formation at Jbel Issoumour, Morocco; holotype complete specimen, IRSNB a1713, close-up of cranidium in plain view. B. Kettneraspis elliptica (Burmeister, 1843) from the Ahrdorf Formation in Gees, Germany; complete specimen, SMF 88192 (=SMF 102k), close-up of cranidium in dorsal view. Length (sag.) of both cranidia is approximately 8 mm.
5Family Odontopleuridae Burmeister, 1843
6Subfamily Odontopleurinae Burmeister, 1843
7Discussion. Ramsköld & Chatterton (1991) revised Leonaspis Richter & Richter, 1917 and assigned the majority of its species to Kettneraspis Prantl & Přibyl, 1949 and their new genus Exallaspis (an acidaspidine). Kettneraspis was considered to be pandemic whereas Leonaspis seemed to be exclusively peri-Gondwanan but new finds of Avalonian origin indicate that the latter did in fact cross the Rheic Ocean during the Emsian (e.g. Basse in Basse & Müller, 2004; Feist & McNamara, 2007; van Viersen, 2013). Although the polyphyly of Leonaspis had been largely addressed by Ramsköld & Chatterton (1991), an investigation on the ingroup structure of Kettneraspis was beyond the scope of that paper. Adrain & Ramsköld (1997) transferred a number of Kettneraspis species to their new genus Edgecombeaspis and discussed potential apomorphies of the former while underlining the need for a comprehensive analysis. One of these potential apomorphies is the anterior “pinching out” of the eye ridge: this ridge is almost fully deflated anteriorly just before it reaches the median glabellar lobe as if a deep, longitudinal furrow separates the two (e.g., Plate 1B). In most odontopleurines including Leonaspis, the eye ridge is connected anteriorly to the median glabellar lobe without any interruption.
8In the past two decades a wealth of odontopleurine material was discovered in Devonian strata in the Ardenno-Rhenish Mountains and commercially mined sections in Morocco. Probably the most widely accepted of Ramsköld & Chatterton’s (1991) characters to discriminate between Leonaspis and Kettneraspis has been the number of interior pygidial border spines: four in the first genus as against two in the latter (e.g., Plate 1F). No subsequent worker appears to have had any noteworthy problem with assigning species to either genus as long as the pygidium was known (e.g. Kim, 1997; Basse, 1998; Feist & Orth, 2000; Sandford, 2000; Ebach, 2002; Basse in Basse & Müller, 2004; Chatterton & Ludvigsen, 2004; Basse & Franke, 2006; Chatterton et al., 2006; Feist & McNamara, 2007; van Viersen, 2007, 2008, 2013; Basse & Lemke, 2008; Franke, 2010; Carvalho, 2012). Kennedy (1994) repeated the old, polyphyletic concept of Leonaspis, probably not having had the opportunity to take note of Ramsköld & Chatterton’s (1991) work.
9We argue here that the use of pygidial spines as a key feature in distinguishing between these two genera is not without controversy. The reason for this is that we regard some species as being intermediate in bearing putative synapomorphies of both clades. To illustrate our case three taxa from the Devonian of Morocco are considered, all of which are known from published complete specimens. The first, Leonaspis haddanei, was described in the Timrhanrhart Formation (Emsian) by Chatterton et al. (2006). It has four interior pygidial border spines but it also bears features characteristic of Kettneraspis, including 12-16 cheek border spines, a row of 20 tubercles on the anterior cranidial border, and relatively few, clearly defined and similarly spaced epiborder tubercles (see Ramsköld & Chatterton, 1991). Leonaspis haddanei furthermore has the anteriorly deflated eye ridge, a potential apomorphy of Kettneraspis as alluded to above. The second species is Kettneraspis? prescheri sp. nov. It has two interior pygidial border spines but also eyes set far posteriorly, opposite the occipital furrow, and a narrow fixigenal field. These last two features were held by Ramsköld & Chatterton (1991) to be characteristic of Leonaspis. The third species is represented by a complete specimen of imprecise stratigraphic and geographic origin (“Devonian of Morocco”) described by Basse & Lemke (2008) as Kettneraspis n. sp. 3. It has two interior pygidial border spines but also bears typical Leonaspis features such as eyes set far posteriorly, numerous epiborder tubercles and a narrow fixigenal field. The row of eight long spines on the anterior border of K. n. sp. 3 is unprecedented and likely autapomorphic. Similar but shorter spines, however, have been reported in other species such as Kettneraspis hollowayi Sandford, 2000 and considered to be of paedomorphic origin (Sandford, 2000).
10We find it hard to accept that the above named similarities are all mere convergences on the opposite genus. In fact, without a pygidium available, L. haddanei would best have been assigned to Kettneraspis. It could be postulated that this species is indeed a Kettneraspis with a Leonaspis-type pygidial spine pattern, which is not entirely inconceivable since the four-spined state is a plesiomorphy that would require a “simple” reversal to attain. Potential evidence in support of this suggestion comes from a unique pygidium of the oldest known Kettneraspis species, K. jaanussoni (Chatterton & Perry, 1983), which has two pairs of partially fused interior pygidial border spine pairs (Ramsköld & Chatterton, 1991, p. 337, fig. 3e). Ramsköld & Chatterton, who considered the four-spined condition to be an atavism, suggested that amalgamation may have led to the reduced spines in Kettneraspis. The number of pygidial interior border spines may also have a bearing on the enigmatic origin of Leonaspis. Ramsköld & Chatterton did not deem a derivation of Leonaspis from Kettneraspis likely because that would have required a reversal of two pairs of interior pygidial border spines. Such a derivation may be easier to accept in light of the new data.
11The goal of the present work is not to perform a timely revision of Kettneraspis nor does the generic assignment of K.? prescheri sp. nov. based on its pygidial spine pattern infer our acceptance of this character as being discriminative from Leonaspis. We contemplated on transferring L. haddanei to Kettneraspis but considered this would be a pointless act as long as other species are being ignored. According to Basse in Basse & Müller (2004) the diversity of Leonaspis in Germany is significant and may warrant further subdivision of this genus. Additionally, none of the Avalonian taxa from Germany were recognised at the time of Ramsköld & Chatterton’s (1991) work and these were obviously not included in their analysis. The putative monophyly of Leonaspis may need to be reappraised.
12Genus Kettneraspis Prantl & Přibyl, 1949
13Type species. Acidaspis pigra Barrande, 1872, from the Acanthopyge Limestone (Eifelian) of Konĕprusy, Czech Republic, by original designation.
14Remarks. This is the first potential Kettneraspis species formally described from Morocco. Alberti (1969, 1970) and Feist & Orth (2000) recorded specimens from the Eifelian which they identified as Kettneraspis pigra. Chatterton et al. (2006) provided a list of Leonaspis species from Morocco.
15Kettneraspis? prescheri sp. nov.
16(Fig. 2A, Pl. 1, Figs A-F, Pl. 2, Figs A, B)
17Leonaspis sp.; Hartl & Winkler Prins, 2001, p. 12, 13, fig. 6c.
18Etymology. In memory of Harald Prescher.
19Holotype. IRSNB a1713, complete specimen.
20Type locality and horizon. “Couche rouge”, Ihandar Formation at Jbel Issoumour, Morocco.
21Diagnosis. Comparatively scantily ornamented Kettneraspis species with a large hook-shaped, distally slightly laterally tapered and blunted median occipital spine occupying almost entire sagittal length of occipital ring and carrying a tubercle on its dorsoventrally highest point. Narrow, border-parallel strip of closely spaced pits just adaxial to librigenal border furrow. Thoracic posterior pleural bands each bear a single short, digitiform dorsal spine abaxially – otherwise devoid of prosopon. Pygidium forms a homogeneous horizontal plane, the axial lobe and pleural ridges excepted.
22Description. Anterior cranidial margin transverse (dorsal view), bearing 12 laterally expanded tubercles, rarely alternated with slightly more ventrally positioned smaller ones. Anterior border dorsally flattened, mildly concave (anterior view), devoid of ornament. Border furrow not developed in front of median glabellar lobe. Posterior border bears a single tubercle distally; maximum length (exsag.) is three times minimum length. Posterior border furrow is a transverse groove that disappears just abaxially to visual surface. Ocular sutures divergent from ω before flexing inward and running towards ε which is obscured by the eye (dorsal view); path ε–δ–γ depicts a high parabola (tr.); pre-ocular sutures exsagittally running toward β opposite L2, convergent between β and α but less so from lateral to anterior margin of median glabellar lobe. Occipital ring with steep lateral sides (anterior view); slightly longer medially (sag.); devoid of ornament. S0 only developed lateral to median glabellar lobe. Small palpebral lobe, bearing a central pit which is adaxially surrounded by a single row of small tubercles. Eye ridge progressively convergent anteriorly, bearing three pairs of tubercles, proximally weakly inflated but well demarcated by groove-like axial furrow, rudimentary opposite S1, well demarcated anteriorly by deep grooves on both sides, weakly inflated anteriorly near median glabellar lobe. L1 tongue-shaped, length axis directed at a 20-25° angle from sagittal line, devoid of prosopon. S1 shallow to rudimentary. L2 teardrop-shaped, bearing a few tubercles. S2 deep. L3 not developed. Longitudinal glabellar furrow is rudimentary except near lateral glabellar furrows. Median glabellar lobe with a low-concave anterior margin, angular main body (plain view), depicting a quarter circle (lateral view), slightly laterally expanded anteriorly, bearing four tubercle pairs, first opposite posterior half of L1, second opposite S1, third opposite S2, and fourth just anterolateral to third; tubercles of each consecutive pair positioned more apart; exsagittal row of smaller tubercles on both sides of median glabellar lobe; apart from this basic pattern there may be a few randomly scattered tubercles.
23Librigenal border broad, progressively tapered anteriorly. Epiborder tubercles above border spines 4, 7, 9 and 11. There are 14 border spines the adaxialmost of which (i.e., those in front of the eyes) have laterally expanded tips; towards posterior each spine is longer and more acuminate. Border furrow is shallow except near genal angle, generally mostly recognisable by the change of slope between border and librigenal field. 60-70° angle between lateral border furrow and posterior border furrow (lateral and dorsal views). Librigenal field is most inflated anteriorly; width is 0.75 of length (dorsal view); ornament consists of a few closely spaced tubercles anteriorly under the eye; otherwise smooth. Eye is tall, conical, much higher than occipital ring, placed opposite S0. Very long, thin genal spines that diverge at a close to 80° angle proximally and subsequently very gently converge, bearing very fine, randomly scattered granules; two dorsal spines on the base of the genal spine (character 17:2 of Ramsköld & Chatterton, 1991), these being a short thorn opposite second thoracic axial ring and a granule between the former and ω.
24Thorax consists of nine segments. Axis convexly rounded (posterior view), tapered (tr.) progressively posteriorly; width of ninth axial ring relative to first is 0.6:1. Axial rings slightly W-shaped, each bearing a single, spiny tubercle pair but less pronounced on anterior segments (no evidence found of a serially homologous pair on occipital ring); otherwise devoid of ornament. Anterior pleural bands of each segment are about as long (tr.) as axial ring. The anterior two posterior pleural bands bear a single tubercle abaxially and short, spiny lateral extremities. Segments 3 to 9 macropleural; posterior pleural bands weakly vaulted; posterior pleural spine pair of third segment is longest, longer than genal spine but also proximally slightly thinner and more convergent than the latter, mildly tapered throughout; remaining posterior pleural spines backward flexed and considerably tapered proximally, from there resembling a (straight) needle. Pleural furrow of each segment is a shallow groove.
25Pygidium broadly rounded, about three times wider than long, carrying a tubercle pair positioned anterolaterally. First axial ring deeply (sag.) incised posteriorly by a small pseudoarticulating half ring. Second axial ring bears a single tubercle pair. Terminal axial piece of similar size to pseudoarticulating half ring. Pleural ridge is proximally posterolaterally oriented, then flexed posteriorly, bears a single tubercle just posterior to flexure; distally inward curved. Two interior border spines between slightly convergent major border spines. Single tubercle present centrally on the base of both major border spines. Three, possibly four, pairs of lateral border spines.
26Remarks. The adaxialmost librigenal border spines are not preserved on the left cheek in the holotype. The right cheek is slightly displaced and its adaxial extremity lies underneath the cranidium. Based on other specimens in private collections that were examined by us 14 border spines could be corroborated.
27There is a single tubercle on top of the occipital spine in K.? prescheri sp. nov. which is opened in the holotype, meaning that it was damaged or perhaps formed the basis of an organ that was not preserved. In most of the specimens that we have examined the tubercle is barely recognisable (usually effaced during preparation) but always found in the same place.
28The number of pygidial lateral border spines is variable between Kettneraspis species. It is not clear whether there is a fourth spine pair in K.? prescheri sp. nov. This pair can easily be concealed by the adjacent thorax segment as previously noticed in congeners by Bruton (1968).
29Paedomorphosis. Kettneraspis? prescheri sp. nov. demonstrates a number of features of possibly paedomorphic origin. None of the specimens studied by us are juveniles. The resemblances of some of these characters to known juvenile stages of Kettneraspis are nonetheless worth mentioning.
30Paired axial glabellar tubercles 2, 3, 4 and 5 are invariably present on all the specimens that we have seen. One specimen of Kettneraspis elliptica (Burmeister, 1843) from Gees (Rhenish Mountains, Germany), also has them although the anteriormost pair is barely visible in dorsal view (Fig. 2B, Plate 2C). We did not see paired tubercles on several other individuals of K. elliptica that were examined by us. This is perhaps not surprising as most trilobites from the Gees site (famous “Trilobitenfelder”) were collected over a period of roughly two centuries, under limited or no stratigraphical control (van Viersen et al., 2009). Thus, it is conceivable that they came from very different stratigraphic levels. Other species of Kettneraspis, Leonaspis and Edgecombeaspis may also show paired tubercles (see, e.g., Alberti, 1969, pl. 40, figs 14a, 15; Chatterton & Perry, 1983, pl. 10, fig. 11, pl. 13, fig. 4) but they appear to be easily obscured by the randomly scattered tubercles on the glabella (see, e.g., Alberti, 1969, pl. 40, fig. 10a; Haas, 1969, pl. 84, fig. 1c). Paired axial glabellar tubercles are already present in the earliest of odontopleurids from the Cambrian (see Bruton, 1983, p. 882, pl. 88, figs 12, 15). They are probably homologous with spines shown by juvenile stages of some Kettneraspis species (see, e.g., Chatterton, 1971, pl. 8, fig. 19).
31The convex occipital ring (anterior view) and long median spine of our new species remind of meraspid Kettneraspis cranidia figured by Chatterton (1971, p. 44, fig. 11B) and Whittington & Campbell (1967, pl. 16, figs 9, 10, 12-14). In large holaspides of Kettneraspis species the occipital ring is usually lower and the median spine rudimentary. As van Viersen (2007) pointed out, there are few Kettneraspis species known that bear a well developed occipital spine. Kettneraspis bayarti van Viersen, 2007 from the Jemelle Formation (Eifelian) in Belgium has a slender but long, backward curved spine. A more closely similar example to our species, however, is the occipital spine of Kettneraspis favonia (Haas, 1969) from the Rabbit Hill Limestone (Pragian) of Nevada (US) although that is somewhat thinner and shorter, if not broken off.
32Comparisons. Beside the similar occipital ring and median spine, K.? prescheri sp. nov. and K. favonia share a number of other features such as the shallow lateral border furrow, the ornament on librigenal field restricted to area underneath the eye, the shallow S0 posterior to median glabellar lobe and the short spine distally on at least some of the thoracic posterior pleural bands. Although both species are likely closely related, Haas’ species is principally different in having deeper S1, two well-developed (long) dorsal spines on the base of the genal spine, and numerous tubercles on the eye ridge, occipital ring and pygidium.
33Specimens from the Pragian in Bohemia assigned to Leonaspis ruderalis (Hawle & Corda, 1847) by Přibyl & Vaněk (1966, pl. 1, figs 5-7) include a cranidium with an affinity to our species in having a large occipital spine and S0 weakly developed posterior to the median glabellar lobe. The librigena and pygidium recorded by Přibyl & Vaněk are clearly different in bearing numerous epiborder tubercles and four interior border spines, respectively.
34Leonaspisissoumourensis was described by Alberti (1970) based on a single pygidium which is presumably from the Emsian at Jbel Issoumour, the type locality of K.? prescheri sp. nov., and thus should be discussed. Alberti’s pygidium differs in generally showing much more relief with in particular an inflated posterior border, and in having four interior border spines.
4. Acknowledgements
35K. Weddige and M. Basse allowed AV to examine and photograph specimens in the collections of the Senckenberg Institute in 2008. F. Lerouge (Leuven) participated in helpful discussions. The manuscript benefited from review comments provided by M. Basse and A. Bignon.
5. References
36Adrain, J.M. & Ramsköld, L., 1997. Silurian Odontopleurinae (Trilobita) from the Cape Phillips Formation, Arctic Canada. Journal of Paleontology, 71, 237-261.
37Alberti, G. K. B., 1969. Trilobiten des jüngeren Siluriums sowie des Unter- und Mitteldevons. I. Mit Beiträgen zur Silur-Devon Stratigraphie einiger Gebieten Marokkos und Oberfrankens. Abhandlungen der Senckenbergischen Naturforschenden Gesellschaft, 520, 1-692.
38Alberti, G. K. B., 1970. Trilobiten des jüngeren Siluriums sowie des Unter- und Mitteldevons. II. Abhandlungen der Senckenbergischen Naturforschenden Gesellschaft, 525, 1-233.
39Barrande, J., 1872. Systême Silurien du centre de la Bohême. Ière partie. Recherches paléontologiques, I: Trilobites, Crustacés divers et Poissons (Supplément). Prague and Paris, xxx + 647 p.
40Basse, M., 1998. Trilobiten aus mittlerem Devon des Rhenohercynikums: III. Proetida (3), Phacopida (2), Lichida (Lichoidea, Odontopleuroidea) und ergänzende Daten. Palaeontographica (A), 249, 1-162.
41Basse, M. & Franke, C., 2006. Marine Faunen aus dem frühen Unteremsium (Unterdevon) des Givonne-Oesling-Antiklinoriums (Luxemburg). Ferrantia, 46, 7-41.
42Basse, M. & Lemke, U., 2008. Drei neue Trilobiten aus dem Devon von Marokko. Der Aufschluss, 59, 173-180.
43Basse, M. & Müller, P., 2004. Eifel-Trilobiten III. Corynexochida, Proetida (2), Harpetida, Phacopida (2), Lichida. Quelle & Meyer-Verlag, Wiebelsheim, 261 p.
44Bruton, D.L., 1968. A revision of the Odontopleuridae (Trilobita) from the Palaeozoic of Bohemia. Skrifter utgitt av Det Norske Videnskaps-Akademi i Oslo. I. Matematisk-naturvidenskapelig Klasse. Ny Serie, 25, 1-73.
45Bruton, D.L., 1983. Cambrian origins of the odontopleurid trilobites. Palaeontology, 26, 875-885.
46Burmeister, H., 1843. Die Organisation der Trilobiten aus ihren lebenden Verwandten entwickelt; nebst einer systematischen Übersicht aller seither beschriebenen Arten. Reimer, Berlin, 147 p.
47Carvalho, M. da G.P. de, 2012. Trilobites. In Racheboeuf, P.R., Casier, J.-G., Plusquellec, Y. Toro, M., Mendoza, D., Carvalho, M. da G.P. de, Le Hérissé, A., Paris, F., Fernández-Martínez, E., Tourneur, F., Broutin, J., Crasquin, S. & Janvier, P., New data on the Silurian-Devonian palaeontology and biostratigraphy of Bolivia. Bulletin of Geosciences, 87, 299-301.
48Chatterton, B.D.E., 1971. Taxonomy and ontogeny of Siluro-Devonian trilobites from near Yass, New South Wales. Palaeontographica (A), 137, 1-108.
49Chatterton, B.D.E., Fortey, R.A., Brett, K.D., Gibb, S.L. & McKellar, R.C., 2006. Trilobites from the upper Lower to Middle Devonian Timrhanrhart Formation, Jbel Gara el Zguilma, southern Morocco. Palaeontographica Canadiana, 25, 1-177.
50Chatterton, B.D.E. & Ludvigsen, R., 2004. Early Silurian trilobites of Anticosti Island, Québec, Canada. Palaeontographica Canadiana, 22, 1-264.
51Chatterton, B.D.E. & Perry, D.G., 1983. Silicified Silurian odontopleurid trilobites from the Mackenzie Mountains. Palaeontographica Canadiana, 1, 1-127.
52Ebach, M.C., 2002. Lower Devonian trilobites from Cobar, New South Wales. Records of the Western Australian Museum, 20, 353-378.
53Feist, R. & McNamara, K.J., 2007. Biodiversity, distribution and patterns of extinction of the last odontopleuroid trilobites during the Devonian (Givetian, Frasnian). Geological Magazine, 144, 777-796.
54Feist, R. & Orth, B., 2000. Trilobites de la limite Eifélien/Givétien de la région stratotypique (Tafilalet, Maider, Maroc). In Tahiri, A. & El Hassani, A. (eds), Proceedings of the Subcommission on Devonian Stratigraphy (SDS) – IGCP 421 Morocco Meeting. Travaux de l’Institut Scientifique Rabat, Série Géologie & Géographie Physique, 20, 78-91.
55Franke, C., 2010. Marine Fauna der Wiltz-Schichten (Ober-Emsium, Unter-Devon) der Mulde von Wiltz und der Daleider Mulden-Gruppe (Luxemburg, Deutschland): Teil 1. Ferrantia, 58, 5-62.
56Haas, W., 1969. Lower Devonian trilobites from central Nevada and northern Mexico. Journal of Paleontology, 43, 641-659.
57Hartl, F. & Winkler Prins, C., 2001. Trilobieten in vogelvlucht: de collectie van het Nationaal Natuurhistorisch Museum Naturalis. Gea, 34 (3), 10-16.
58Hawle, I. & Corda, A.J.C., 1847. Prodrom einer Monographie der böhmischen Trilobiten. Calve, Prague, 176 p.
59Kennedy, R.J., 1994. British Devonian trilobites. Part 1. Monograph of the Palaeontological Society, 148, 1-33.
60Kim, I.-S., 1997. Trilobiten aus dem Greifensteiner Kalk (Grenzbereich Unter-Mittel Devon). Palaeontographica (A), 245, 157-205.
61Morzadec, P., 2001. Les Trilobites Asteropyginae du Dévonien de l'Anti-Atlas (Maroc). Palaeontographica (A), 262, 53-85.
62Prantl, F. & Přibyl, A., 1949. Studie o trilobitech nadčeledi Odontopleuracea nov. superfam. Rozpravy státního geologického ústavu ČSR, 12, 1-221. [1-110 in Czech, 111-115 in Russian, 117-221 in English]
63Přibyl, A. & Vaněk, J., 1966. Zur Kenntnis der Odontopleuridae-Trilobiten aus dem böhmischen Altpaläozoikum. Acta Universitatis Carolinae – Geologica, 4, 289-304.
64Ramsköld, L. & Chatterton, B.D.E., 1991. Revision and subdivision of the polyphyletic 'Leonaspis' (Trilobita). Transactions of the Royal Society of Edinburgh: Earth Sciences, 82, 333-371.
65Richter, R. & Richter, E., 1917. Über die Einteilung der Familie Acidaspidae und über einige ihrer devonischen Vertreter. Centralblatt für Mineralogie, Geologie und Paläontologie, 1917, 462-472.
66Sandford, A.C., 2000. Silurian trilobite faunas and palaeoenvironmental setting of the early Ludlow Melbourne Formation, central Victoria. Alcheringa, 24, 153-206.
67Viersen, A.P. van, 2007. Kettneraspis, Radiaspis and Ceratarges (Trilobita) from the Middle Devonian of the Rochefort area (Ardennes, Belgium). Scripta Geologica, 134, 1-18.
68Viersen, A.P. van, 2008. First record of the odontopleurid trilobite Koneprusia from the lower Eifelian of Vireux-Molhain (northern France), with remarks on the associated fauna. Geologica Belgica, 11, 83-91.
69Viersen, A.P. van, 2013. Latest Early to early Middle Devonian acastid trilobites from the eastern part of the Dinant Synclinorium, Belgium (Rhenohercynian Zone). Memoirs of the Association of Australasian Palaeontologists, 44, 1-10.
70Viersen, A.P. van, Prescher, H. & Savelsbergh, J., 2009. Description of two new trilobites from the Ahrdorf Formation (Middle Devonian) at the “Trilobitenfelder” of Gees, Eifel, Rhenish Mountains. Bulletin de l’Institut royal des Sciences naturelles de Belgique, Sciences de la Terre, 79, 43-53.
71Whittington, H.B., 1956. Silicified Middle Ordovician trilobites: The Odontopleuridae. Bulletin of the Museum of Comparative Zoology, 114, 155-288.
72Whittington, H.B. & Campbell, K.S.W. 1967. Silicified Silurian trilobites from Maine. Bulletin of the Museum of Comparative Zoology, 135, 447-482.
73Whittington, H.B. & Kelly, S.R.A., 1997. Morphological terms applied to Trilobita. In Kaesler, R.L. (ed.), Treatise on invertebrate paleontology, Part O, Arthropoda 1. Trilobita, revised. Volume 1: Introduction, Order Agnostida, Order Redlichiida. Geological Society of America, Boulder and University of Kansas Press, Lawrence, 313-329.
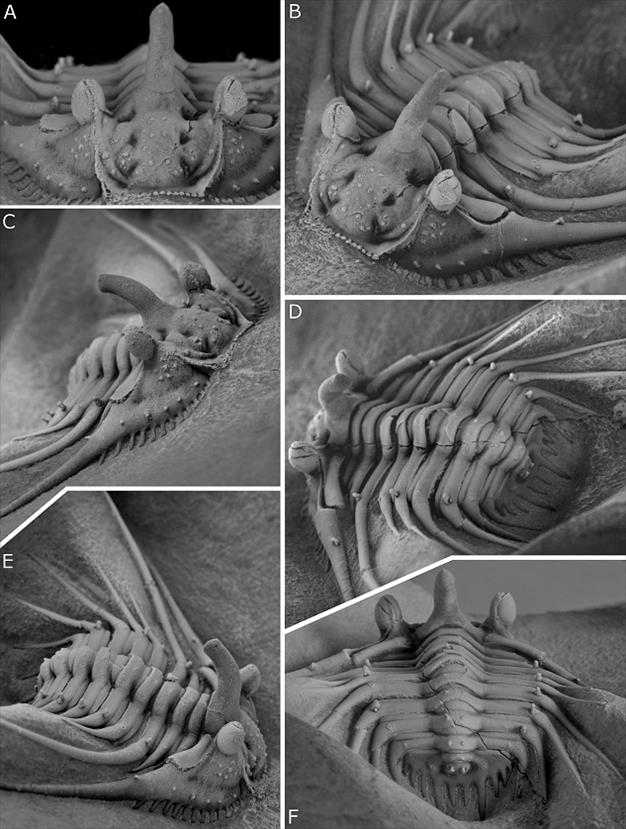
Plate 1. Kettneraspis? prescheri sp. nov. from the Ihandar Formation at Jbel Issoumour, Morocco.
A-F. holotype complete specimen, IRSNB a1713, in anterior (A), oblique anterior (B), anterolateral (C), oblique posterior (D), oblique lateral (E) and posterior (F) views. Specimen is 17 mm wide (tr.) across fourth thorax segment (excluding spines).
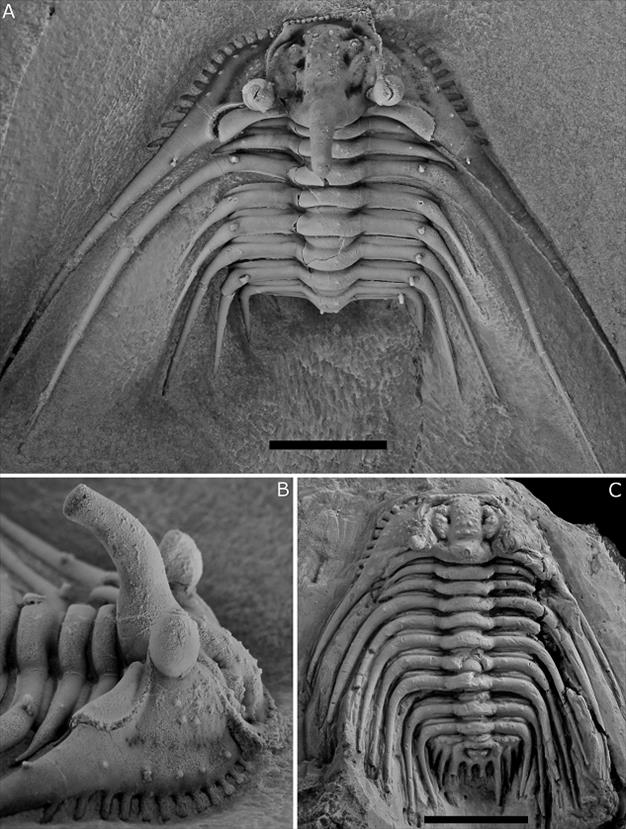
Plate 2. Kettneraspis? prescheri sp. nov. from the Ihandar Formation at Jbel Issoumour, Morocco.
A, B. holotype complete specimen, IRSNB a1713, cephalothorax in dorsal view (A) and close-up of cephalon in lateral view (B).
Kettneraspis elliptica (Burmeister, 1843) from the Ahrdorf Formation in Gees, Germany.
C. complete specimen, SMF 88192 (=SMF 102k), in dorsal view.
Scale bars represent 10 mm.
74Manuscript received 15.10.2014, accepted in revised form 17.12.2014, available on line 14.01.2015.