- Accueil
- volume 16 (2012)
- numéro 2
- Optimization of water potential and nutrient levels for Kentucky bluegrass – white clover mixture on acidic soils
Visualisation(s): 4003 (52 ULiège)
Téléchargement(s): 95 (1 ULiège)
Optimization of water potential and nutrient levels for Kentucky bluegrass – white clover mixture on acidic soils

Notes de la rédaction
Received on November 8, 2010; accepted on June 27, 2011
Résumé
Optimisation du potentiel hydrique et du niveau des éléments nutritifs des prairies à pâturin – trèfle blanc sur sols acides. Dans les prairies des Appalaches, l'acidité du sol et la topographie très variée sont les principaux facteurs qui affectent la productivité. Il est nécessaire de développer une fonction de réponse pour le rendement et de déterminer la valeur nutritive des prairies. Les facteurs du sol, comme le potentiel hydrique (WP), le pH, la teneur en azote (N) et en phosphore (P) sont très variables à travers les prairies de montagne. Une étude en serre a été menée afin d'étudier les effets de ces différents facteurs sur la production en herbe et la concentration en nutriments des prairies mixtes à pâturin des prés (Poa pratensis L.) et trèfle blanc (Trifolium repens L.). Un dispositif composite central rotatif a été utilisé pour mener une étude en pots. Les effets de deux niveaux de WP et de cinq niveaux de chacun des facteurs pH, N et P ont été évalués. Les facteurs WP, pH, N et P et leurs interactions WP x pH, WP x N, pH x N, et N x P expliquent une part importante de l'accumulation de l'herbe dans notre essai. L'influence des différents facteurs sur les variations d'accumulation de l'herbe est, dans l'ordre décroissant : WP > pH > WP x pH > WP x N > P > N > N x P > pH x N. D'après nos expérimentations, les conditions optimales pour la production du système de culture pâturin – trèfle blanc sont les suivantes : WP de -422 à -171 kPa, pH du sol de 5,5 à 6,1 ; teneur en N de 50 à 68 mg·kg-1, et teneur en P de 38 à 40 mg·kg-1. Les concentration (%) en azote (N), phosphore (P), potassium (K), calcium (Ca) et magnésium (Mg) ont été déterminées dans les tissus des tiges pour comprendre l'impact des différents facteurs sur la composition nutritive du fourrage. Le WP et le pH du sol ont montré une influence significative sur les concentrations de tous les éléments. Le pH du sol et la teneur en N ont un effet significatif sur la concentration en N dans les tissus de la plante. La concentration en P est significativement influencée par l'interaction teneur en P avec le WP et le pH du sol. La fonction de réponse du rendement du mélange pâturin des prés et trèfle blanc de cette étude devrait être évaluée en champ pour obtenir une meilleure fiabilité des résultats. Les effets du WP, du pH, des teneurs en N et P sur l'accumulation de l'herbe et sa valeur nutritive doivent être mieux compris pour améliorer la productivité dans un paysage vallonné.
Abstract
In Appalachian grasslands, soil acidity and highly variable topography are the main factors negatively affecting the productivity. There was a need to develop a yield response function and determine nutritive value of grasslands. Soil factors such as water potential (WP), soil pH, nitrogen (N) and phosphorus (P) levels are highly variable across mountainous grasslands. A greenhouse study was conducted to study the effects of WP, soil pH, N, and P levels on the herbage production and nutrient concentration of Kentucky bluegrass (Poa pratensis L.) and white clover (Trifolium repens L.) mixed sward. Central composite rotatable design was used to conduct this pot study. The effects of two levels of WP and five levels each of pH, N, and P fertilizers were evaluated. WP, pH, N and P levels as their interactions WP x pH, WP x N, pH x N, and N x P explained a significant part of sward accumulation in our trial. The importance of the different factors in explaining herbage accumulation variations was, in decreasing order, WP > pH > WP x pH > WP x N > P > N > N x P > pH x N. Optimum conditions for the production of bluegrass and white clover crop system were predicted, from this pot experiment, as follows WP of -422 to -171 kPa, 5.5 to 6.1 soil pH, 50 to 68 N mg·kg-1, and 38 to 40 P mg·kg-1. Concentration (%) of nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), and magnesium (Mg) were determined in shoot tissue to understand the impact of the different factors on nutrient content of forage. WP and soil pH had shown significant influence on concentration of all elements. Effects of soil pH and N level had significant effect on N concentration in plant tissue. Plant P concentration was significantly influenced by interaction of P level with WP and soil pH. Yield response function of Kentucky bluegrass and white clover mixture from this case study should be evaluated in field trials to obtain practical significance. Effect of WP, soil pH, and N and P levels on herbage accumulation of grasslands and nutritive value should be understood to harmonize productivity across this undulating landscape.
Table des matières
1. Introduction
1Grassland productivity of mountainous Appalachian region (West Virginia) has been affected due to topographic, soil, and climatic constraints. Grazing is the most important economic activity of any mountainous grassland requiring conservation, enhancement of productivity, and adaptation of biodiversity (Watkinson et al., 2001). The high precipitation in these mountainous grasslands of Appalachian region can lead to leaching of cations such as Ca2+ and Mg2+ and cause the soil exchangeable complex to become dominated by H+ and Al3+ (Ritchey et al., 2002). Dominance of H+ and Al3+ ions in soil exchangeable complex causes acidity which limits crop growth and utilization of many essential nutrients by plants (Haynes et al., 1981; Black, 1993). Topography of mountainous landscape is also highly variable, influencing the distribution of soil water, organic matter, nutrients, soil texture and structure, soil depth and other soil properties that affect plant growth within a field (Zebarth et al., 1989; Pennock et al., 1994). Spatial variability in crop yield is directly correlated with the spatial variability of soil properties across the landscape. Understanding the spatial variability of soil is critical to develop appropriate soil management and precision farming techniques in order to harmonize the crop productivity across the landscape (Bing et al., 2004).
2Soils of recently reclaimed acid grasslands may have imbalance in the concentration of cations and anions due to liming and addition of fertilizers. Remediation of soil acidity is crucial for improving grassland soil quality and increasing forage yield. Liming to remediate acidic soils has a longer history than any other forms of amendments (McLean, 1971). The accuracy of lime recommendation depends on the quality of correlation and calibration of chemical buffer methods for the soils of that particular region (Rajesh et al., 2010). Liming results in changes in chemical and physical properties of soil that improve conditions for plant growth (Menzies et al., 1994). Soil water content that varies with topography is a predominant phenomenon in undulating grasslands. There is a need to understand the interactions of soil water content on the availability of plant essential nutrients to determine the precise agronomic practices to promote on acidic grasslands.
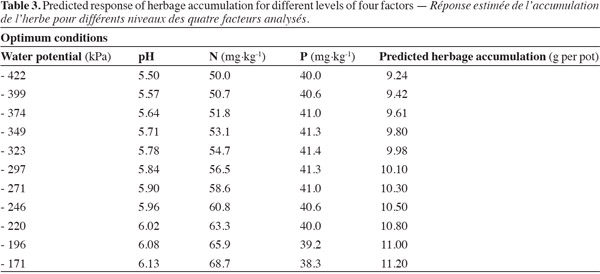
3Legumes are an important component to be incorporated in acid reclaimed grasslands and have been shown to increase herbage dry matter yield and quality (Rhodes et al., 1997; Malhi et al., 2002). Most important biological N2-fixing process (BNF) is the result of symbiotic relationship between legumes and rhizobia which can fix 12 to 25 Tg N per year in the soil (Herridge et al., 2008). Introduction of legumes in grasslands has an advantage in nitrogen (N) limited systems and reduces the cost of inputs such as mineral N fertilizers (Jarvis et al., 1996). White clover (Trifolium repens L.) is one of the more important legume crops in temperate pastures and can provide acceptably high levels of production under low-input systems (Rochon et al., 2004). It is commonly sown in grasslands of temperate zones around the world to improve fodder quality and to fix atmospheric nitrogen (Ledgard, 2001). Therefore, it is essential to include legumes in acidic pasture lands to maximize productivity of these low-input systems (Hartwig, 1998).
4There is a need to understand the interactions between liming and nutrient levels of soil to achieve higher productive grasslands. Addition of nutrient to soil has an effect on any cropping system only if it is the limiting nutrient for plant growth. Armelin et al. (2007) observed the liming enhanced the dry matter yield of grass forage (Brachiaria decumbens L.) with concomitant application of nitrogen (N) and potassium (K) fertilizers. The liming rate of 2.2 Mg·ha-1 was found optimum to increase forage barley yields, to obtain the soil pH of 5.66, and to increase utilization efficiency of the potassium (K) and phosphorus (P) fertilizers (Michaelson et al., 1987). Application of lime and P-rich fertilizers is essential to raise the productivity of relatively infertile permanent pastures by promoting growth of white clover (Dragomir et al., 2008). The important role of P fertilizers along with lime and N in improving pasture yield and quality was observed in mixed pasture swards (Peoples et al., 1995). Ozgur et al. (2006) found that phosphorus treatments significantly affected dry matter yield and crude protein yield in white clover cultivars. Nitrogen-fixing plants have a greater need for P than nitrate-supplied plants (Israel, 1987). N inputs were recognized as equally important to increase herbage production and provide potential to absorb other essential elements from soil such as potassium (Salon et al., 2001; Collins et al., 2003). Application of lime and fertilizers is essential to restore the productivity of acidic grasslands.
5Soil water content is an essential growth factor that can influence the success of any application of amendments and fertilizers. It has effect on soil pH and elemental speciation of soil solution (Mendoza et al., 1995). Soil water is necessary to release nutrients from applied fertilizers through mineralization reactions (Koerselman et al., 1993). Thus, fertilizer use efficiency is a function of available water (Fiez et al., 1994; Gutierrez-Boem et al., 1998). Saeed et al. (1998) also observed a significant dry matter yield increase of forage sorghum (Sorghum bicolor L.) in response to irrigation treatments. In this study, the irrigation treatments were frequent (8 mm per day for every 7 days), intermediate (8 mm per day for every 10 days) and infrequent (8 mm per day for every 13 days), irrigation regimes led to dry matter yields of 16.3, 11.8, 10.5 Mg·ha-1 respectively. Oomes et al. (1997) observed an increase of water level from -30 cm to -5 cm in grassland caused a decrease in N uptake (from 14.1 to 11.4 g N per m2). But both above dry matter production (566 to 690 g per m2) and root mass (82 to 363 g DM per m2) increased by rise of water level. Positive linear relationship in between soil water and phosphorus uptake has been observed (Olsen et al., 1961). Phosphorus uptake by wheat was enhanced by soil water in low P available soils (Strong et al., 1980). Generally, the pasture soils are highly heterogeneous in terms of soil water and nutrient contents due to variability of topography (Noorbakhsh et al., 2008). Soil water also can be influenced by fluctuations in rainfall and air temperatures that ultimately affect the soil solution chemistry (Litaor, 1988). There is need to understand the effects of variable soil and nutrient factors like soil water potential, soil pH, nitrogen and phosphorus on the productivity of acidic grassland which helps to obtain knowledge of precise agricultural practices for mountainous grasslands.
6This greenhouse study was designed to derive yield response model for legume-based pastures and also to quantify the interaction effects of water potential (WP), soil pH, nitrogen (N), and phosphorus (P) levels on Kentucky bluegrass and white clover cropping system.
2. Materials and methods
7Acidic soil (0-15 cm) was collected to use it as medium for pots from Westmoreland soil map unit (Westmoreland fine loamy, mixed, active, mesic ultic hapludalfs) (NRCS, 1999) which belongs to Alfisol. The soil texture of the pot medium was a clay loam (34.3% clay; 44.0% silt), with an initial pH of 4.8, 9.2% organic matter, 42.5 mg·kg-1 calcium (Ca), 9.8 mg·kg-1 magnesium (Mg), 40.8 mg·kg-1 potassium (K), 4.06 mg·kg-1 phosphorus (P), and cation exchange capacity (CEC) of 15.84 cmolc·kg-1. Soil was air-dried, crushed and passed through a 2 mm sieve. Soil pH (1:1) was determined by glass membrane electrode; texture by the pipette method (Sternberg et al., 1961); organic matter content by loss on ignition (Oliver et al., 2001); and cation exchange capacity (CEC) by the ammonium acetate method (Chapman, 1965). Direct calcium hydroxide (Ca(OH)2) titration (Alabi et al., 1986) was done to determine the lime response curve for soil. The soil water characteristic curve was determined using a dew-point potentiometer (Decagon Model WP4-T) and gravimetry. All determinations were done to samples of three replications.
8Based on the lime response curve, sufficient Ca(OH)2 was added to achieve different levels of soil pH ranged from 4.5 to 6.5 in approximately 0.5 increments. Limed soils were subjected to wetting and drying cycles until equilibrium pH was reached. Each pot (30.5 cm x 30.5 cm x 20 cm) was filled with 4 kg of air-dried soil. Five levels of nitrogen (N) and phosphorus (P) fertilizer treatments were applied to the pots. Nitrogen (N) was added at 0, 10, 25, 50 and 100 mg N·kg-1 soil. Phosphorus (P) was added at 0, 10, 20, 40 and 80 mg P·kg-1 soil. Two different water potentials (WP), wet (-50 kPa) and dry (-794 kPa) treatments, as determined from the soil-water characteristic curve, were imposed from 30 days after sowing and consistently maintained using a sensor-based drip irrigation system (NETAFIM Flori 1). Water content was measured weekly by potentiometer (Decagon Model WP4-T) and gravimetric method in soils collected from the pot. Central composite rotatable design (Myers et al., 1995) with three replications was used for this pot study in order to reduce experimental units. Central composite rotatable design is an experimental design to build second order (quadratic) model for the responsible variable without conducting a complete three level factorial experiment. It is a 2k factorial or fraction of 2k factorial design with three groups of design points, including two-level factorial design points, axial or star points, and center points. Effects and interactions between variables or factors were calculated through ordinary least squares method. Pots were directly seeded with mixture of Kentucky bluegrass (Poa pratensis L.) and white clover (Trifolium repens L.) of common varieties. The seed mixture contained 75% bluegrass and 25% white clover (weight basis). The seed rate was 1 g per pot (100 g·100 sq ft-1) with 90% germination percentage. White clover seeds were inoculated with Rhizobium trifoli 2S-2 before sowing. The greenhouse chamber was maintained with air temperature fluctuating between 21-27°C (max) and 16-27°C (min), 50-60% humidity, and 16:8 photoperiod with 230 µE·m-2·s-1 light intensity during the experiment. At the end of the experiment (90 days after germination) herbage dry matter accumulation of bluegrass and white clover mixture (g per pot) was measured by clipping at ground level and drying the herbage at 65°C to constant weight, for 3 days. Nutrient concentration in plants was determined by analyzing the above ground portion in order to understand plant uptake. Dried herbage samples were digested with 70% concentrated nitric acid (HNO3) in microwave (MARS 5, CEM) (Rechcigal et al., 1990). Inductively Coupled Plasma Atomic Emission Spectroscopy (ICP-AES) (Perkin Elmer P400, Norwalk, CT) was used to determine nutrient concentration in wet digested herbage samples. Nutrient concentrations were expressed on a tissue dry mass basis.
9Herbage accumulation data of bluegrass and white clover mixture was analyzed using a response surface methodology with PROC RSREG procedure (SAS version 9.1, SAS Institute, Cary, NC, USA). Herbage accumulation was the response variable and WP, pH, N, and P rates were predictor variables which were used to construct model or response function. Linear, quadratic, and cross product terms of these factors were also determined and used in the multiple regression process. A step-wise regression procedure was used to determine the best model. Adjusted R2 and p-value were used to select model parameters. The effect of WP, soil pH, N, and P levels on the response variable (herbage accumulation) was tested for adequacy and fitness by analysis of variance. Concentrations (%) of nutrients like nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), and magnesium (Mg) in herbage were determined to understand the effect of water potential (WP), pH, N and P levels on plant nutrient content.
3. Results and discussion
3.1. Yield model
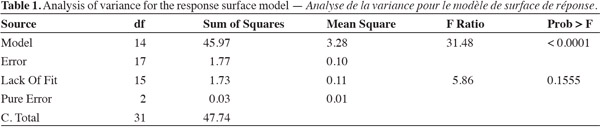
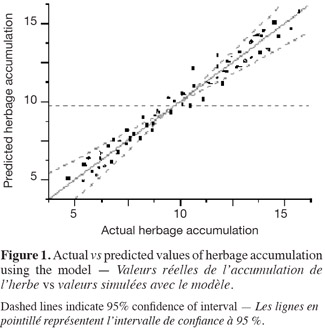
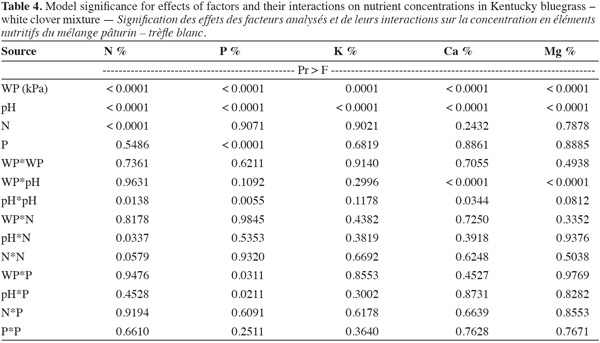
10Analysis of variance showed that the response surface model using factors like water potential (WP), soil pH, nitrogen (N), and phosphorus (P) significantly predicted herbage accumulation (response variable) of bluegrass and white clover mixture (Table 1). The lack of fit term was not significant when mean observational data of experimental units was used. But, high variability in the observational data of experimental units was found due to large effect of treatments. Species composition of bluegrass (75%) and white clover (25%) did not change due to treatments at the time of harvest. Yield response model was validated and observed a significant fit between the observed (actual) yield and predicted yield values of the model (Figure 1). WP, pH, N and P levels were the main effects that influenced herbage accumulation (P < 0.05) (Table 2). Cross product terms such as WP x pH, WP x N, N x P, and pH x N, and also pH x pH (quadratic term) showed significant effect on herbage accumulation.
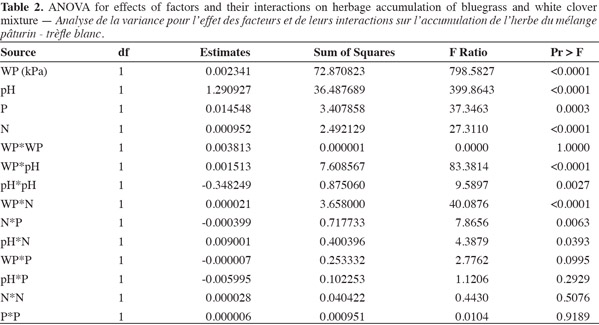
11Water potential (WP) had the largest influence on bluegrass-white clover herbage accumulation, as indicated by sum of squares followed by pH, P and N levels. The order of influence of interaction terms on herbage accumulation was WP x pH > WP x N > N x P > pH x N (Figure 2a-2d). Significant main effects and their interaction terms were used to determine the response surface function to predict herbage accumulation of bluegrass and white clover cropping system:
12Y = (0.002*WP) + (1.29*pH) + (0.009*N) + (0.014*P) + 0.0015*(WP + 422.21)*(pH - 5.5) + 0.00002*(WP + 422.21)*(N - 30.625) - 0.348* (pH - 5.5)*(pH - 5.5) + 0.009* (pH - 5.5)* (N - 30.625) - 0.0003* (N - 30.625)* (P - 25) (Equation 1)
13where Y is herbage accumulation of the bluegrass-white clover mixture in g per pot, WP is water potential in kPa units, N and P are levels of nitrogen and phosphorus supplies in mg·kg-1 respectively.
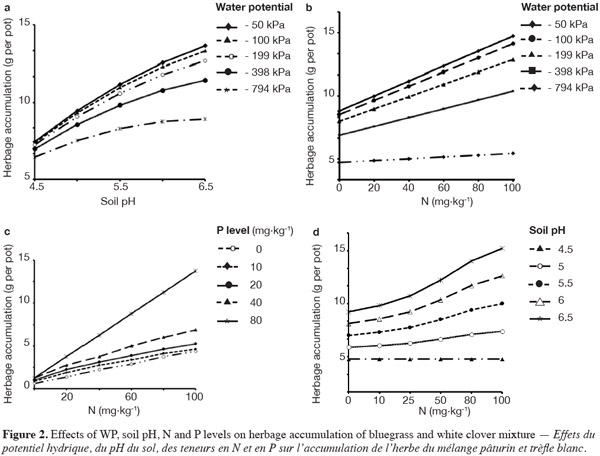
14Parameter estimates of WP, soil pH, N and P levels indicated the positive significant effect on herbage accumulation (Table 2). Liming effect (soil pH) on herbage accumulation was observed as the WP level increased from -794 kPa (Figure 2a). Effects of higher available soil water to plants and liming increased the utilization efficiency of N and P fertilizers that were likely responsible to enhance the herbage accumulation. Liming increased the soil pH from 4.7 to 5.7 and the dry matter yield of annual ryegrass by 4.5 Mg·ha-1 on acid coastal plain soil in Texas (Haby et al., 1996).
15WP by N interaction showed overall significant positive effect on herbage accumulation (g per pot). The rate of increase in herbage accumulation per unit addition of N fertilizer was higher as WP level increased (Figure 2b). At lowest WP level (-794 kPa), the rate of increase in herbage accumulation for increased N fertilizer was lower than for higher WP levels. Increase of water potential in soil was assumed to enhance the herbage accumulation by increasing the N fertilizer use efficiency and herbage accumulation. Labuschagne et al. (2006) investigated the response of perennial ryegrass (Lolium perenne L.) and white clover (Triflium repens L.) to four N rates ranges from 0 to 180 kg N·ha-1 at four soil water potentials of -10 to -35 kPa. Primary dry matter production of ryegrass was responded positively to increase of N (0-180 kg N·ha-1) and soil water potential levels (-10 to -35 kPa) and recorded with higher yields. But white clover were not influenced by N rate and soil water potential. Abassi et al. (2005) observed higher dry matter and N uptake in grass swards with addition of N at below field capacity moisture level (71% water filled pore space). So the sufficient water potential is necessary to make soil nutrients available to plants (fertilizer use efficiency) through processes such as dissolution and hydrolysis (Fiez et al., 1994).
16The interaction between N and P levels also showed a statistically significant effect on herbage accumulation (g per pot) (Figure 2c). As the rate of applied N and P fertilizers increased, the herbage accumulation also showed a significant positive response. In this study, herbage accumulation response to N supply was stronger at the highest rate of P (80 mg·kg-1) fertilizer. Application of N and P fertilizers seemed to have synergistic effect on herbage accumulation by increasing plant height, higher number of leaves per plant, and greater leaf area. Celebi et al. (2011) also observed the significant positive effect of N and P fertilizer on green herbage, hay and crude protein of natural pastures. Herbage accumulation (g per pot) also responded significantly to the rate of nitrogen (N) along with rise in soil pH due to liming (Figure 2d). As the pH increased from 5.5 to 6.5 with the increase in N rate, herbage accumulation also increased. The response of herbage accumulation was highest as soil pH increased from 4.5 to 5.0. But there was a decrease in response of herbage accumulation to N additions at pH above 5.0. Liming seemed to increase N availability by increasing the uptake of applied N fertilizer which helped the growth of bluegrass and white clover mixture. Armelin et al. (2007) observed the increase in dry matter yield and mineral content of degraded pasture with Brachiaria decumbens due to liming (4 t·ha-1) and application of N fertilizer.
17In this study, the optimum levels of WP, pH, N, and P were determined to maximize herbage accumulation of bluegrass and white clover mixture (Table 3). Higher herbage accumulation (g per pot) can be achieved at water potential (WP) ranged from -171 kPa to -422 kPa, pH from 5.50 to 6.13, N from 50 to 68.7 mg·kg-1, and P from 38.3 to 40 mg·kg-1.
3.2. Nutrients concentrations in bluegrass – white clover herbage
18The impact of WP, soil pH, N, and P and their interaction effects on nutrient concentration of bluegrass and white clover mixture were determined in order to understand the plant nutrient uptake. This study also helps to obtain knowledge for establishing the fertilizer application management based on nutrient cycling in bluegrass and white clover mixture. Concentrations (%) of nutrients such as nitrogen, phosphorus, potassium, calcium, and magnesium were determined (Table 4). WP and pH had significant effects on the concentration of all nutrients in plant tissue (Figure 3a-3e). In previous studies, liming had shown significant positive effect on uptake of P, S, Ca, Mg by wheat (Sultana et al., 2009) and also increased the concentration of Ca, Mg, and P in mountain Norway spruce needles (Kulhavy et al., 2009).
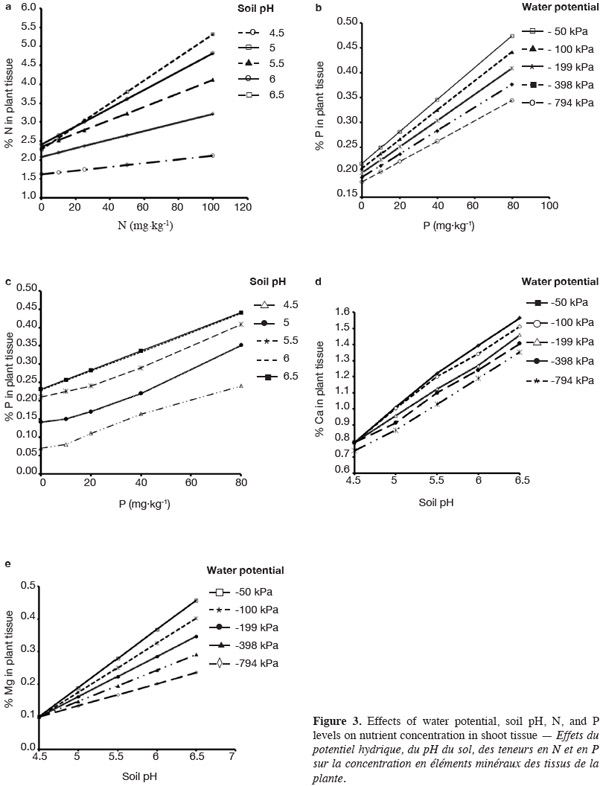
19N concentration (%) in herbage samples was significantly affected by WP, pH and N fertilizer rate. N concentration (%) in plant tissue samples of different treatments ranged from 1.62 to 5.32 with standard deviation (SD) of 0.94. Highest plant N concentration was observed at high water potential (-50 kPa) with 100 mg N·kg-1 and soil pH 6.5. Maximum response (with highest slope) in N concentration per unit N fertilizer applied was observed as soil pH increased from 4.5 to 5.0 (Figure 3a). Liming and high water consumption seemed to increase the mineralization of applied N fertilizer and uptake by plants. Stevens et al. (1996) also observed the increase of N uptake as the lime (0 to 8 t·ha-1) and N fertilizer (80 to 160 kg N·ha-1 per year) rates increased in two sward types (permanent pasture and perennial ryegrass reseed).
20Phosphorus concentration (%) was significantly affected by WP, pH, and P level. P concentrations (%) in the plant samples from all treatments were varied from 0.18 to 0.47 with standard deviation (SD) of 0.08. The increased of P concentration in plant tissue at the higher WP level may be due to enhancement in the diffusion process (Olsen et al., 1961) (Figure 3b). Increase in pH up to 6.0 also had a significant effect on P concentration (Figure 3c). But P concentration was not affected by the increase of soil pH above 6.0. There was a reduction in response of plant P concentration as soil pH increased above 5.0. Liming increases the availability of P to plants (up to soil pH below 9.0) by dissolution of aluminum and iron phosphates (Bardgett, 2005). Nuttall (1980) also measured a significant increase of P concentration in alfalfa (Medicago media) and bromgrass (Bromus inermis) from 0.16 to 0.30% with the application of P fertilizer (20 kg·ha-1). Concentration of K, Ca, and Mg in shoots was significantly affected by WP and pH of soil (Table 4). K, Ca, and Mg in plant tissue samples were ranged from 1.31-3.48 (SD = 0.61), 0.70-1.56 (SD = 0.25), and 0.09-0.45 (SD = 0.10) respectively. WP and pH showed significant effect on the concentration of Ca and Mg in herbage samples (Figure 3d-3e). The increased concentrations of K, Ca, and Mg were observed at higher WP levels, possibly due to an increase in the magnitude of mobilization processes such as mass flow and diffusion (Mackay et al., 1985). In this study, the increase in pH by liming also enhanced the concentration of cations (K, Ca, and Mg) in plant tissue. Liming reduces the toxic effect of H+ and Al3+ in acidic soils and enhances the root growth and cation uptake by plants (Bolan et al., 2003; Tang et al., 2003; Malavolta, 2006).
4. Conclusion
21Soil acidity is the major limiting factor which affects negatively the dry matter yield and nutrient concentration of forages in Appalachian region. In this region, highly variable topography causes large variation in physical and chemical properties of soil which ultimately results the variation in productivity across the landscape. This greenhouse study was conducted with the treatments similar to the conditions of Appalachian pastures in which the soil properties such as water potential, soil acidity, and N, and P levels vary across the landscape. In this case study, the results showed that WP, soil pH, and N and P levels had significant effects on dry matter yield of bluegrass and white clover system and its nutrient concentration. Yield response function was derived for bluegrass and white clover system using soil factors such as water potential, soil pH, N, and P levels. Optimization of WP, pH, N, and P levels were done to maximize the herbage production of bluegrass and white clover mixture. Nutrient concentration (%) in shoot tissue was determined in this study, because the mineral nutrition represents the uptake, transport, metabolism, and utilization of nutrients by any cropping system. A specific minimum amount of all nutrients is necessary for healthy forage growth and its determination also could help to establish fertilizer application management for grasslands. WP, soil pH and its interaction had shown significant effect on N, P, K, Ca, and Mg concentration in shoot tissue. Increase in nutrient concentration was mainly associated with increased dry matter yields. The positive effect of liming in this greenhouse study is therefore not surprising; liming seemed to improve the exploitation of applied fertilizers and soil water and made nutrients available for plant uptake. The similar impacts of water potential, liming and N, and P level were observed in bluegrass cropping system (Rajesh et al., 2012). Further field trials with similar treatments are to be conducted to evaluate the yield response function of bluegrass and white clover mixture and impact of WP, soil pH, N and P rates on nutrient concentrations in plant tissue. There is also a need to corroborate the optimization of water potential, soil pH, N, and P levels for bluegrass and white clover mixture in field conditions. Yield response function for a specific cropping system is a very useful tool to design accurate management strategies to harmonize the production across the highly heterogeneous landscape.
22Acknowledgements
23Scientific contribution number 3048 from the West Virginia Agricultural and Forestry Experiment Station, Morgantown, WV. Funding for this research was provided from the Pasture-Based Beef Production for Appalachia project, a multi-institutional collaboration among West Virginia University, USDA-ARS, Virginia Tech, and University of Georgia and from funds appropriated under the Hatch Act.
Bibliographie
Abassi K., Mushtaq K. & Fayyaz U.H., 2005. Nitrogen use efficiency and herbage production of an established grass sward in relation to moisture and nitrogen fertilization. J. Plant Nutr., 28, 1693-1708.
Alabi H.E., Sorensen R.C., Knudsen D. & Rehm G.W., 1986. Comparison of several lime requirement methods on coarse textured soils of northeastern Nebraska. Soil Sci. Soc. Am. J., 50, 937-947.
Armelin M., Primavesi O., Primavesi A. & Saiki M., 2007. Effect of liming and fertilizer on mineral content and productivity of Brachiaria decumbens grass forage. J. Radioanal. Nucl. Chem., 271, 221-224.
Bardgett R.D., 2005. The biology of soil. Oxford, UK: Oxford University Press.
Bing C.S. & Ferrell R.E., 2004. Scale dependent relationship between wheat yield and topographic indices: a wavelet approach. Soil Sci. Soc. Am. J., 68, 577-587.
Black A.C., 1993. Soil fertility evaluation and control. Boca Raton, FL, USA: Lewis Publishers.
Bolan N.S., Adriano D.C. & Curtin D., 2003. Soil acidification and liming interactions with nutrient and heavy metal transformation and bioavailability. Adv. Agron., 78, 215-258.
Celebi S.Z., Arvas O. & Terzioglu O., 2011. The effect of nitrogen and phosphorus fertilizer application on herbage yield of natural pastures. Pak. J. Biol. Sci., 14, 53-58.
Chapman H.D., 1965. Cation exchange capacity. In: Black C.A., ed. Methods of soil analysis. Part 2: Chemical and microbiological properties. Madison, WI, USA: American Society of Agronomy, 891-900.
Collins R.P., Fothergill M., Maduff J.H. & Puzio S., 2003. Morphological compatibility of white clover and perennial ryegrass cultivars grown under two nitrate levels in flowing solution culture. Ann. Bot., 92, 247-258.
Dragomir N. et al., 2008. White clover's contribution (Trifolium repens L.) to the increase of yield and quality of temporary hil pastures. Agriculture, 65, 78-83.
Fiez T.E., Miller B.C. & Pan W.L., 1994. Winter wheat yield and grain protein across varied landscape positions. Agron. J., 86, 1026-1032.
Gutierrez-Boem F.H. & Thomas G.W., 1998. Phosphorus nutrition affects wheat response to water deficits. Agron. J., 90, 166-171.
Haby V.A., Hillard J.B. & Clary G., 1996. Liming acid soils for ryegrass production. Better Crops, 80, 14-15.
Hartwig U.A., 1998. The regulation of symbiotic N2 fixation: a conceptual model of N feedback from the ecosystem to the gene expression level. Perspect. Plant Ecol., 1, 92-120.
Haynes R.J. & Ludecke T.E., 1981. Effect of lime and phosphorous applications on concentrations of available nutrients and on P, Al, and Mn uptake by two pasture legumes in an acid soil. Plant Soil, 62, 177-188.
Herridge D.F., Peoples M.B. & Boddey R.M., 2008. Global inputs of biological nitrogen fixation in agricultural systems. Plant Soil, 311, 1-18.
Israel D.W., 1987. Investigation of the role of phosphorus in symbiotic dinitrogen fixation. Plant Physiol., 84, 835-840.
Jarvis S.C., Wikins R.J. & Pain B.F., 1996. Opportunities for reducing the environmental impact of dairy farming managements: a systems approach. Grass Forage Sci., 51, 21-31.
Koerselman W., Van Kerkhoven M.B. & Verhoeven J.T.A., 1993. Release of inorganic N, P and K in peat soils; effect of temperature, water level. Biogeochem., 20, 63-81.
Kulhavy J., Markova I., Drapelova I. & Truparova S., 2009. The effect of liming on the mineral nutrition of the mountain Norway spruce (Piecea abies L.) forest. J. For. Sci., 55, 1-8.
Labuschagne J. & Agenbag G.A., 2006. The effect of fertilizer N rates on growth of perennial ryegrass (Lolium perenne) and white clover (Trifolium repens) grown at high soil water levels under controlled conditions. S. Afr. J. Plant Soil, 23, 215-224.
Ledgard S.F., 2001. Nitrogen cycling in low input legume-based agriculture, with emphasis on legumes/grass pastures. Plant Soil, 228, 43-59.
Litaor M.I., 1988. Soil solution chemistry in an alpine watershed, Front Range, Colarado, USA. Arct. Alp. Res., 20, 485-491.
Mackay A.D. & Barber S.A., 1985. Soil moisture effects on root growth and phosphorus uptake by corn. Agron. J., 77, 519-523.
Malavolta E., 2006. Manual of mineral nutrition of plants. Piracicaba, Brazil: Editora Ceres.
Malhi S.S., Zentner R.P. & Heier K., 2002. Effectiveness of alfalfa in reducing fertilizer N input for optimum forage yield, protein concentration, returns and energy performance of bromegrass-alfalfa mixtures. Nutr. Cycling Agroecosyst., 62, 219-227.
McLean E.O., 1971. Potentially beneficial effects from liming: chemical and physical. Soil Crop Sci. Soc. Fla. Proc., 31, 189-196.
Mendoza R.E., Collantes M.B., Anchorena J. & Cazinzos S., 1995. Effects of liming and fertilization of forage yield and vegetation in dry heath soils from Tierra del Fuego. J. Plant Nutr., 18, 401-420.
Menzies N.W., Bell L.C. & Edwards D.G., 1994. Exchange and solution phase chemistry of acid, highly weathered soils. 1. Characteristics of soils and effect of lime and gypsum amendments. Aust. J. Soil Res., 32, 251-267.
Michaelson G.J. & Ping C.L., 1987. Effects of P, K, and liming on soil pH, Al, Mn, K, and forage barley dry matter yield and quality for a newly-cleared Cryorthod. Plant Soil, 104, 155-161.
Myers R.H. & Montgomery D.C., 1995. Response surface methodology: process and production in optimization using designed experiments. New York, USA: John Wiley & Sons, Inc.
Noorbakhsh S., Schoenau J., Zeleke B.S.T. & Qian P., 2008. Soil properties, yield, and landscape relationships in south-central Saskatchevan Canada. J. Plant Nutr., 31, 539-556.
NRCS, 1999. Soil taxonomy, a basic system of soil classification for making and interpreting soil surveys. 2nd ed. Washington, DC, USA: USDA-NRCS.
Nuttall W.F., 1980. Effect of nitrogen and phosphorus fertilizers on a bromegrass and alfalfa mixture grown under two systems of pasture management. II. Nitrogen and phosphorus uptake and concentration in herbage. Agron. J., 72, 295-298.
Oliver H., Lotter A.F. & Lemcke G., 2001. Loss on ignition as a method for estimating organic and carbonate content in sediments: reproducibility and comparability of results. J. Paleolimnol., 25, 101-110.
Olsen S.R., Watanabe F.S. & Danielson R.E., 1961. Phosphorus absorption by corn roots as affected by moisture and phosphorus concentration. Proc. Soil Sci. Soc. Am., 25, 289-294.
Oomes M.J.M., Kuikman P.J. & Jacobs F.H.H., 1997. Nitrogen availability and uptake by grassland in mecocosms at two water levels and two water qualities. Plant Soil, 192, 249-259.
Ozgur T. & Sebahattin A., 2006. Effects of different levels of phosphorus on forage yield and quality of white clover (Trifolium repens L.). Asian J. Plant Sci., 5, 201-206.
Pennock D.J., Anderson D.W. & de Jong E., 1994. Landscape-scale changes in indicators of soil quality due to cultivation in Saskatchewan, Canada. Geoderma, 64, 1-19.
Peoples M.B. et al., 1995. Effects of surface application of lime and superphosphate to acid soils on growth and N2 fixation by subterranean clover in mixed pasture swards. Soil Biol. Biochem., 27, 663-671.
Rajesh C., McDonald L.M. & Bryan W.B., 2010. Grouping of soils by taxonomic order to improve lime recommendations. Commun. Soil Sci. Plant Anal., 41, 1594-1603.
Rajesh C., McDonald L.M. & Bryan W.B., 2012. Effect of soil water and nutrients on productivity of Kentucky bluegrass system in acidic soils. J. Plant Nutr., 35, 288-303.
Rechcigal R.W. & Payne G.G., 1990. Comparison of microwave digestion system to other digestion methods for plant tissue analysis. Commun. Soil Sci. Plant Anal., 21, 2209-2218.
Rhodes I. & Ortega F., 1997. Forage legumes. In: Weddell J.R., ed. Seeds of Progress. British Grassland Society Occasional Symposium No. 31. Reading, UK: British Grassland Society, 15-27.
Ritchey K.D. & Snuffer J.D., 2002. Limestone, gypsum and magnesium oxide influence the restoration of an abandoned Appalachian pasture. Agron. J., 94, 830-839.
Rochon J.J. et al., 2004. Grazing legumes in Europe: a review of their status, management, benefits, research needs and future prospects. Grass Forage Sci., 59, 197-214.
Salon C. et al., 2001. Grain legume seed filling in relation to nitrogen acquisition: a review and prospects with particular reference to pea. Agronomie, 21, 539-552.
Saeed I.A.M. & El-Nadi A.H., 1998. Forage sorghum yield and water use efficiency under variable irrigation. Irrig. Sci., 18, 67-71.
Sternberg R.W. & Creager J.S., 1961. Comparative efficiencies of size analysis by hydrometer and pipette methods. J. Sediment. Petrol., 31, 96-100.
Stevens R.J. & Laughlin R., 1996. Effects of lime and nitrogen fertilizer on two sward types over a 10-year period. J. Agric. Sci., 127, 451-461.
Strong W.M. & Barry G., 1980. The availability of soil and fertilizer phosphorus to wheat and rape at different water regimes. Aust. J. Soil Res., 18, 353-362.
Sultana B.S. et al., 2009. Effect of liming on soil properties, yield and nutrient uptake by wheat. Curr. World Environ., 4, 39-47.
Tang C., Rengel Z., Diatloff E. & Grazey C., 2003. Responses of wheat and barley to liming on a sandy soil with subsoil acidity. Field Crops Res., 80, 235-244.
Watkinson A.R. & Ormerod S.J., 2001. Grassland, grazing and biodiversity: editor's introduction. J. Appl. Ecol., 38, 233-237.
Zebarth B.J. & de Jong E., 1989. Water flow in a hummocky landscape in Central Saskatchewan, Canada, I. Distribution of water and soils. J. Hydrol., 107, 309-327.
Pour citer cet article
A propos de : Rajesh Chintala
South Dakota State University. North Central Sun Grant Center. 815 Medary Avenue. Suite 201. 57006 Brookings, South Dakota (USA). E-mail: rajesh.chintala@sdstate.edu
A propos de : Louis M. McDonald
West Virginia University. Division of Plant and Soil Sciences. 1090 Agricultural Sciences Bldg. P.O. Box 6108. Morgantown, WV 26506-6108 (USA).
A propos de : William B. Bryan
West Virginia University. Division of Plant and Soil Sciences. 1090 Agricultural Sciences Bldg. P.O. Box 6108. Morgantown, WV 26506-6108 (USA).





