- Accueil
- Volume 15 (2011)
- numéro spécial 1
- An improved protein extraction method for detecting ruminant material in feed using lateral flow device dipsticks
Visualisation(s): 3287 (10 ULiège)
Téléchargement(s): 48 (1 ULiège)
An improved protein extraction method for detecting ruminant material in feed using lateral flow device dipsticks

Abstract
The currently used immunological lateral flow based dipstick technology is not sensitive to detect the presence of ruminant animal material below 1% level of contamination in feed. The possible reason is that the processed animal proteins are subjected to chemicals and to high temperatures during the rendering process, thereby leading to either their denaturation or degradation. Also some feed samples producing viscous extracts or containing high contents of mineral or vitamins delay the migration flow in the test device and may thus give rise to either inconclusive or false positive results. Veterinary Laboratory Agency focused its efforts on developing a new protein extraction method through the EU Safeed-PAP project with the aim to increase the sensitivity of the dipstick method. The method developed is based on a bone sedimentation procedure coupled with an EDTA digestion step in order to liberate the protected proteins which are covalently bound to mineral structures within the bone mineral cage. All the results using this method showed that the feed contaminated with ruminant material is detected at 0.1 to 0.2% levels.
Table des matières
1. Introduction
1Since the outbreak of Bovine Spongiform Encephalopathy (BSE) and the naming of meat and bone meal (MBM) as the main vector of the spread of the disease, the use of processed animal proteins (PAP) in the manufacture of feed has been banned in the European Community (EC). A number of European national projects have been undertaken in order to develop suitable methods to detect the presence of mammalian MBM in compound feeds. Lack of specific analytical methods on high heat treated meat and bone meal have led from mammalian ban to a total animal products ban. EC decision 2000/766 (European Commission, 2000) and EC regulation 999/2001 (European Commission, 2001) stated that Member States shall prohibit the feeding of:
2– proteins derived from animals to ruminants;
3– processed animal proteins to farmed animals which are kept, fattened or bred for the production of food.
4In order to improve the impact of the total ban, European Commission requires development of new tests which would differentiate animal proteins at species level. This would help European Commission in considering the relaxation of methods in adding certain species specific proteins to the animal feed.
5The immunological lateral flow device kit Reveal Ruminant of the Neogen corporation presents a great advantage in handling and screening samples for presence of ruminant material. However, some feed samples producing viscous extracts or samples which had high mineral or vitamin contents have shown delayed migration and produced inconclusive results. The sensitivity of the kit is currently at 2% level. Veterinary Laboratory Agency, UK has taken up the responsibility to study under Safeed project work package-2 to improve the species-specific detection of animal proteins in animal feed, to improve the sensitivity levels and to reduce false positive and false negative results. During the rendering process, MBM is subjected to chemicals and to high temperatures and the animal proteins present are either denatured or degraded. Therefore, to extract detectable native proteins, a method was developed which is based on a bone sedimentation procedure coupled with an EDTA digestion step. In this process the proteins which are covalently bound to mineral structures within the bone mineral cage are protected from high temperature and chemical damages are available for detection using different methods.
2. Materials and methods
6The approach used was aimed at complete breakdown of bone fragments into their mineral and protein constituents. It is known that the proteins are covalently bound to mineral structures and the mineral cage of the bone could potentially protect the protein molecules during the rendering process. Therefore, the new extraction procedure focused on recovering undamaged proteins present within the mineral cage which are extracted by decalcifying the bones with EDTA free acid. The main advantage of this extraction procedure is that the extracts can be used for different analytical methods as the collected proteins were not subjected to major chemical or temperature insults.
2.1. Samples
7Negative feed matrix. Five hundred grams each of sunflower, maize, rape seed, wheat and soya ingredients were combined to produce 2,500 g of composite negative feed matrix. The ingredients and composite matrix were tested and confirmed for absence of animal material using microscopic analysis, ELISA (using antibodies against heat stable proteins) and PCR methods (animal mitochondrial 16s RNA gene).
8Meat and bone meal. Pure meat and bone meals from various species were obtained from a pilot plant where the samples were treated at 133°C, 137°C, 141°C, 145°C and at minimum 3 bars for at least 20 min according to the EU legislation.
9Test sample preparation. Positive samples were prepared individually by spiking from 0.1% to 5% pure species specific MBM samples to the negative feed matrix. To prepare 0.5% contaminated sample: 38.8 g of negative feed matrix individually weighed into a container and contaminated with 0.2 g (0.5%) of a pure species specific rendered meat and bone meal.
2.2. Protein extraction from sedimented bone
10Sedimentation. Using EU approved MAT (Microscopic Analysis Test) method [European Commission Directive (EC) No 152/2009]: Weigh 20 (± 1) g of each sample into a pre-weighed sedimentation glass and add 100 (± 5) ml of tetra chloro ethylene (TCE) to it in a fume hood chamber. Stir thoroughly for 30 s with a wooden applicator/stick and leave it for 3 min. At the end of 3 min stir once again for 30 s and leave it for another 3 min. Pour out the vegetable matter carefully by pouring out the float and TCE in one continuous motion (without losing the sediment).
11Wash the sediment. To get rid of soluble minerals and vitamins: add 15 (± 1) ml of water to the sediment, stir thoroughly for 30 s with a wooden applicator/stick and leave it for 3 min. At the end of 3 min, pour out the water without losing sediment.
12Remove phospholipids and other lipid contents. Add 15 (± 1) ml of acetone and n-butanol (1:1) mixture to the sediment, stir thoroughly with a wooden applicator/stick for 1 min and leave it for 5 min. Pour out the organic layer and retain the sediment in the sedimentation glass (avoid loss of the sediment during washings).
13Dry the sediment completely in a hot air oven ~ at 70°C (3 h).
14Bone digestion. To the known weight of dry sediment (~ 200 to 500 mg) add three times the weight of free EDTA acid (e.g. for 300 mg of sediment add 900 mg of EDTA free acid). To this mixture add 2 ml of deionised water to every 100 mg of sediment taken and shake it overnight for at least 16-18 h on a shaker or rotor at 160 RPM (rotations per minute). Following day to the digested bone paste add 4 ml of PBS (pH 7) and mix gently.
15Adjust the pH of the extract in the range between 7.5 -8.0 using pH strips. Dip the pH paper in the extract and read the pH. If the pH is below 7 adjust the pH range to 7.5 to 8.0 using 20 N NaOH. If the pH range is above 8 use 5N HCl. Alternatively pH meter can be used to adjust the pH (if pH meter is used, care is to be taken to avoid sample contamination: each time clean the probe thoroughly between samples). Allow the mixture to settle for 15 min and collect the supernatant into pre-labeled Eppendorf tubes.
16Dilutions and test. Prepare another set of Eppendorf tubes and dilute the supernatants 1:4 distilled water and incubate the tubes at 50°C for 10 min. Test the warmed up extract with Reveal for Ruminant in Feed dipsticks (Neogen Corporation, USA) for 10 min. While testing, continue to keep the Eppendorf tubes on a thermal plate at 50°C.
17Interpretation of results obtained with the Neogen dipstick assay Reveal for Ruminant in Feed. If a line forms in the test zone and other line forms in control zone within 10 min (2 lines in total), the sample is positive. A positive sample may be determined as soon as 2 lines are visible on the strip. If there is no line in the control zone, the test is invalid and the sample should be retested with another strip. If after the full 10 min there is no visible line in the test zone but a visible line in the control zone, the sample is negative. Observations after 15 min may be inaccurate due to overdevelopment of the device. To avoid overdevelopment after 10 min, and retain the test results snip off the solid red bottom portion of the strip to stop the wicking of sample extract through the test and control zone.
18If a prozone effect is suspected (Butch, 2000) prepare another 2 sets of Eppendorf tubes and dilute the supernatants 1:6, 1:8 with distilled water and incubate the tubes at 50°C for 10 min. Test the warmed up extract with Reveal for Ruminant in Feed dipsticks (Neogen Corporation, USA) for 10 min. The positive results upon further dilution confirms the prozone effect.
3. Results
19The results presented in table 1 show that pure processed animal proteins (PAP) rendered at different temperatures were detected correctly. Proteins extracted from bone sediments rendered up to 145°C and diluted 1:4 were correctly identified on all the ruminant samples. This shows that the proteins covalently bound to bone mineral structures are protected from denaturation or fragmentation.
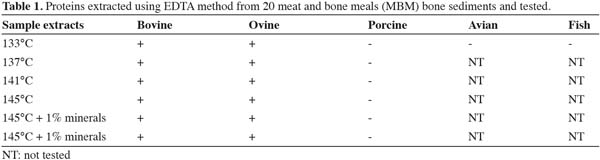
20Furthermore, a set of eight feed samples spiked with 0.1%, 0.2%, 0.3%, 0.4%, 0.5% bovine MBM along with 1% porcine meal, 1% avian meal and 1% fish meal were tested. Figure 1 reveals that all the ruminant spiked samples even at the content of 0.1% were detected as positive and all the non-ruminant sample extracts showed negative results. The thickness of the band is gradually increased according to the level of contamination showing the sensitivity. The detection limit on Neogen kit is at 2% level whereas our improved method is able to detect at 0.1% level. Once again these results show that the proteins covalently bound to bone mineral structures are protected up to a certain point from denaturation or fragmentation and therefore still detected at low levels.

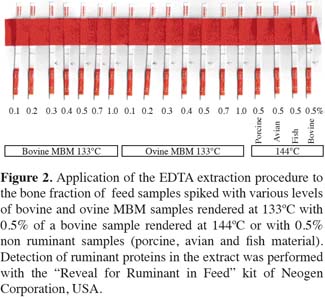
21Another eighteen negative feed matrix samples spiked with 0.1, 0.2, 0.3, 0.4, 0.5, 0.75 and 1% of bovine and ovine PAP rendered at 133ºC and 0.5% bovine sample rendered at 144ºC were tested along with 0.5% porcine, avian and fish meal (non-ruminants) samples. The results in figure 2 show that all the ruminant samples spiked above 0.2% levels were correctly identified with 1:4 dilution.
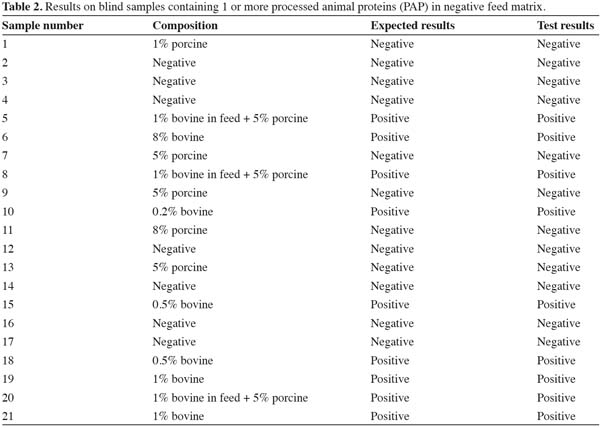
22More tests were carried out on twenty one blind samples to see the ability of the method to detect low levels of more than 1 MBM species in feed and the results are presented in table 2. All the blind samples were correctly identified. The negative feed matrix and non-ruminant samples gave negative results without producing any false positive results. All the ruminant samples were correctly identified in spite of presence of low levels of non-ruminant material in some of the feed samples. These blind sample extracts were also tested with the dipsticks produced under the EU SAFEED-PAP project to compare these results with those obtained when using the Neogen corporation dipsticks. The results were completely identical to what is presented in table 2.
23Further to test the ability of this method for detecting presence of ruminant material in other pure processed animal protein (PAP) (i.e. PAP in PAP. e.g. Bovine MBM in pure porcine/avian/fish meal), 64 samples were tested (data not shown). This method did not produce accurate results on samples containing PAP in PAP. The possible reason could be due to steric hindrance (crowding of nonspecific around specific target protein) on the proximity and orientation of antigen and antibody complex formation.
3.1. Results of interlab studies
24Bone extraction method with free acid EDTA produced the desired results and increased the sensitivity of the dipstick testing from 2% to 0.2%. Therefore, an interlab study was carried out on five samples. Table 3 shows that 3 labs have taken part in the study and all the labs have identified all the positive and negative samples correctly but one lab failed to detect a positive sample. The same sample was correctly identified by the other 2 labs with 1:4 diluted samples.

4. Discussion
25The detectable quality protein extraction approach through bone sediment digestion was aimed at complete separation of bone fragments into their mineral and protein constituents. It is known that the proteins are covalently bonded to mineral structures and the mineral structure of the bone could potentially protect the protein molecules during the rendering process from chemical and temperature insults. Therefore, to recover these proteins a new extraction procedure focused on bone particles by decalcifying the bones with EDTA free acid was developed by VLA. All the results from our laboratory show that the feed contaminated with ruminant material is detected at above 0.1% level. All the tests carried out on bone EDTA extracts suggest that the physical constraints of the mineral cage might prevent protein structures denaturing or fragmenting. Therefore, recovering these unaffected proteins from bone sediments would make it possible to liberate good quality protein material for use as a target in LFD and ELISA methods.
26Acknowledgements
27This study was funded by EU and DEFRA, UK. Special thanks to all the EU consortium members and the staff involved in interlab studies for their cooperation. The author would like to thank all the staff of feed analysis unit and National Feed Audit management of VLA and DEFRA.
Bibliographie
Butch A.W., 2000. Dilution protocols for detection of hook effects/prozone phenomenon. Clin. Chem., 46, 1719-1720.
European Commission, 2000. Council Decision 2000/766/EC of 4 December 2000 concerning certain protection measures with regard to transmissible spongiform encephalopathies and the feeding of animal protein. Off. J. Eur. Union, L306, 07.12.2000, 32-33.
European Commission, 2001. Regulation (EC) No 999/2001 of the European Parliament and of the Council of 22 May 2001 laying down rules for the prevention, control and eradication of certain transmissible spongiform encephalopathies. Off. J. Eur. Union, L147, 31.05.2001, 1-40.
European Commission, 2009. European Commission Regulation (EC) No 152/2009 of 27 January 2009 laying down the methods of sampling and analysis for the official control of feed. Off. J. Eur. Union, L54, 26.02.2009, 1-130
Neogen Corporation Ltd, USA. www.neogen.com
Pour citer cet article
A propos de : Muralidhar Karanam
Veterinary Laboratory Agency (VLA). Department of Environment, Food and Rural Affairs. GB-Luddington, Stratford upon Avon, CV37 9SJ Warwickshire (United Kingdom). E-mail: m.karanam@vla.defra.gsi.gov.uk





